Advertisements
Advertisements
Question
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Advertisements
Solution

Resonance structures of o-nitrophenoxide ion

Resonance structures of p-nitrophenoxide ion
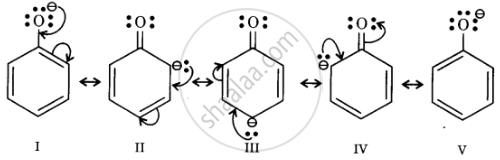
Resonance structures of phenoxide ion
In substituted phenols, electron withdrawing groups such as nitro group increase the acidic strength of phenol. This effect becomes more potent when such groups are present at ortho and para positions. This is because of the effective delocalisation of the anion of the phenoxide ion. Hence, o- and p-nitrophenols are more acidic than phenol.
APPEARS IN
RELATED QUESTIONS
Write the final product(s) in each of the following reactions:

Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to aldehyde.
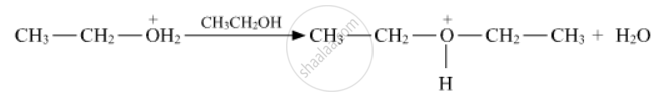
Write the mechanism (using curved arrow notation) of the following reaction :
Which one of the following on oxidation gives a ketone?
The process of converting alkyl halides into alcohols involves ______.
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) |  |
| (b) |  |
| (c) |  |
In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
What is Lucas reagent?
The correct geometry around oxygen in CH3OCH3 is
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
