Advertisements
Advertisements
Question
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
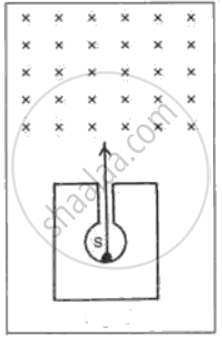
Advertisements
Solution
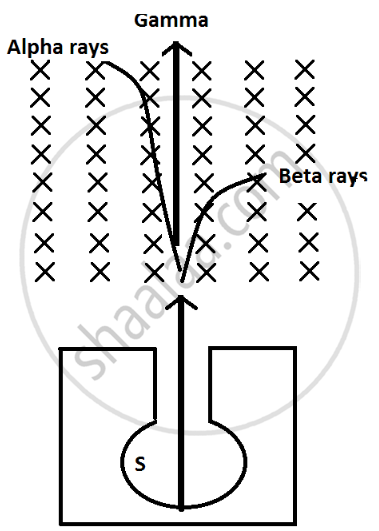
(i) Alpha particles are heavy in mass and are positively charged so they are deflected less by magnetic field and direction is upward which can be calculated by Fleming left hand rule.
(ii) Beta particles are negligible in mass so they are highly deflected by magnetic field and they are negatively charge particle so they are deflected in downward direction.
(iii) Gamma radiations have no mass and no charge so they are not deflected by magnetic field.
APPEARS IN
RELATED QUESTIONS
An element `""_ZS^A` decays to `""_85R^222` after emitting 2 α particles and 1 β particle.
Find the atomic number and atomic mass of the element S.
What is meant by radioactivity?
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
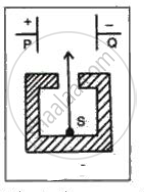
Why is the source S placed in a thick lead container?
A radioactive nucleus `""_"Z"^"A"` X first emits a beta particle and then an alpha particle to give the resulting nucleus `""_"Q"^"P"` Y What will be the values of P and Q in terms of A and Z?
Define bound electrons.
State two characteristics of the substance used as thermionic emitter.
State two properties that a substance should possess when used as a thermionic emitter.
In a cathode ray tube state the purpose of the fluorescent screen.
The activity of a certain radionuclide decreases to 15 percent of its original value in 10 days. What is its half life?
[ln (0.15) = -1.9]
In the following atoms, which one is a radioisotope? Give one use of this isotope.
O16, C14, N14, He4
