Advertisements
Advertisements
Question
In the following atoms, which one is a radioisotope? Give one use of this isotope.
O16, C14, N14, He4
Advertisements
Solution
C14 is a radioisotope. It is used in carbon dating to determine the age of organic materials. It is used in biological research, agriculture, pollution control, and archaeology and so on.
APPEARS IN
RELATED QUESTIONS

Complete the diagram as given above by drawing the deflection of radioactive radiations in an electric field
What do you mean by Atomic mass
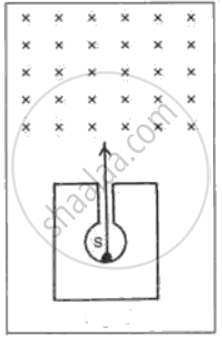
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
State two characteristics of the substance used as thermionic emitter.
State two properties that a substance should possess when used as a thermionic emitter.
In a cathode ray tube state. How is it possible to increase the rate of emission of electrons.
Name the three main parts of a Cathode Ray Tube.
State three common properties of beta rays and cathode rays.
Two radioactive sources A and B of half lives of 1 hour and 2 hours, respectively, initially contain the same number of radioactive atoms. At the end of two hours, their rates of disintegration are in the ratio of ______.
Complete the following radioactive reaction:
\[\ce{^-_-X -> ^-_-Y + ^4_2He -> ^234_91Z + ^0_-1e}\]
