Advertisements
Advertisements
Question
State two characteristics of the substance used as thermionic emitter.
Advertisements
Solution
(i) The substance must have high melting point.
(ii) The work function of the substance should be low so that electrons may be emitted even at low temperature.
RELATED QUESTIONS
Answer the following questions based on a hot cathode ray tube
Name the charged particles
How can they be made to leave the metal surface? (State any two ways)
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
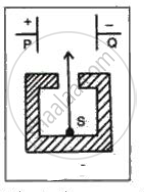
Why is the source S placed in a thick lead container?
A radioactive nuclide `""_86^226` Ra decays by emission of two alpha particles, one beta particle and gamma rays. Which of the following is the resulting nuclide X?
What happens to the position of an element in the periodic table when it emits an alpha particle
A nucleus is \[\ce{^24_11 Na}\] β-radioactive.
Write the equation represent β-decay.
An element P disintegrates by α - -emission and the new element suffers two further disintegrations, both by β -emission, to form an element Q. Explain the fact that P and Q are the isotopes.
What are ‘Becquerel rays’?
Explain, why radium paint, consisting of zinc sulphide and a trace of radium salt, glows in the dark?
Assertion (A): Hydrogen atom consists of only one electron but its emission spectrum has may lines.
Reason (R): Only Lyman series is found in the absorption spectrum of hydrogen atom whereas in the emission spectrum, all the series are found.
