Advertisements
Advertisements
Question
What is meant by radioactivity?
Advertisements
Solution
Elements with a high atomic number such as uranium, thorium, radium have a property of spontaneously emitting invisible, highly penetrating and high energy radiation. This property is called radioactivity. The nucleus of radioactive elements is unstable.
APPEARS IN
RELATED QUESTIONS
Why do Free electrons not leave the metal surface on their own
How can they be made to leave the metal surface? (State any two ways)
A radioactive substance is oxidized. Will there be any change in the nature its radioactivity? Give a reason for your answer
Arrange α, β, and γ rays in ascending order with respect to their
1) Penetrating power.
2) Ionising power
3) Biological effect
Name a gas caused by the Greenhouse effect
What is thermionic emission?
A nucleus `""_11^24Na` emits a beta particle to change into Magnesium (Mg)
(i) Write the symbolic equation for the process.
(ii) What are numbers 24 and 11 called?
(iii) What is the general name of `""_12^24Mg `with respect to `""_11^24Na` ?
Radiations given out from a source when subjected to an electric field in a direction perpendicular to their path are shown below in the diagram. The arrows show the path of the radiation A, B and C. Answer the following questions in terms of A, B and C.

1) Name the radiation B which is unaffected by the electrostatic field.
2) Why does the radiation C deflect more than A?
3) Which among the three causes the least biological damage externally.
4) Name the radiation which is used in carbon dating.
State the penetrating range of α, β and γ radiations.
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
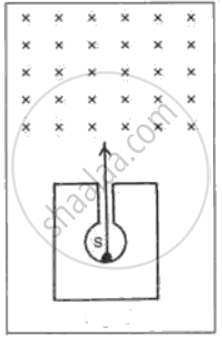
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
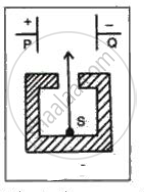
Why is the source S placed in a thick lead container?
A certain nucleus A (mass number 238 and atomic number 92) is radioactive and becomes a nucleus B (mass number 234 and atomic number 90) by the loss of one particle.
What particle was emitted?
An element P disintegrates by α - -emission and the new element suffers two further disintegrations, both by β -emission, to form an element Q. Explain the fact that P and Q are the isotopes.
What are the types of emission?
How is a cathode ray beam affected while passing through a magnetic field?
How is a cathode ray beam affected while passing through an electric field?
What are cathode rays? Why are they called ‘cathode rays’? Name a material which exhibits fluorescence when cathode rays fall on it.
State any two uses of cathode rays.
Justify with reason, whether the following nuclear reactions are allowed or not.
\[\ce{^A_Z X -> ^A_{Z + 1}Y + ^0_{-1}β}\]
Justify with reason, whether the following nuclear reactions are allowed or not.
\[\ce{^A_Z X -> ^A_{Z - 2}X + ^4_{2}He}\]
Which part of the atom undergoes a change in the process of radioactive decay?
State three ways of obtaining an electron-beam.
Mention four properties of Becquerel rays.
Unit of radioactivity is _______
Heavy radioactive elements eventually turn into ______.
The mean lives of a radioactive substance are 1620 and 405 years for β-emission and β-emission respectively. The time after which three fourth of a sample will decay if it is decaying both by β-emission and β-emission simultaneously will be ______ years.
(Take ln 2 = 0.693)
A deuteron strikes 8O16 nucleus with subsequent emission of an alpha particle. Identify the nucleus so produced:
