Advertisements
Advertisements
प्रश्न
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
Advertisements
उत्तर
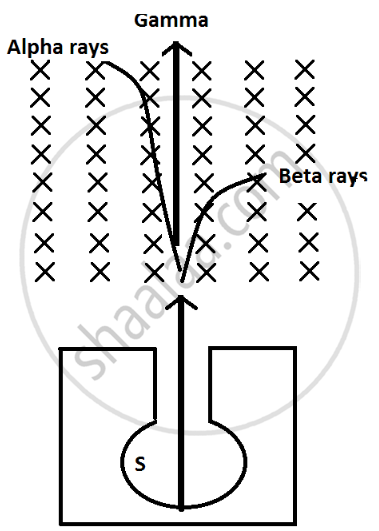
(i) Alpha particles are heavy in mass and are positively charged so they are deflected less by magnetic field and direction is upward which can be calculated by Fleming left hand rule.
(ii) Beta particles are negligible in mass so they are highly deflected by magnetic field and they are negatively charge particle so they are deflected in downward direction.
(iii) Gamma radiations have no mass and no charge so they are not deflected by magnetic field.
APPEARS IN
संबंधित प्रश्न
Why is a cathode ray tube evacuated to a low pressure?
What do you mean by Mass number.
What is nucleus of an atom? Compare its size with that of the atom. Name its constitutents. How is the number of these constituents determined by the atomic number its atomic model.
A radioactive substance is oxidized. What changes would you expect to take place in the nature of radioactivity? Explain your answer.
An element P disintegrates by α - -emission and the new element suffers two further disintegrations, both by β -emission, to form an element Q. Explain the fact that P and Q are the isotopes.
In a cathode ray tube state. How is it possible to increase the rate of emission of electrons.
Which of the following statements is/are correct?
- α particles are photons
- Penetrating power of γ radiation is very low
- Ionization power is maximum for α rays
- Penetrating power of γ radiation is very high
A radioactive element is placed in an evacuated chamber. Then the rate of radioactive decay will ______.
Assertion (A): The force of repulsion between atomic nucleus and α-particle varies with distance according to inverse square law.
Reason (R): Rutherford did α-particles scattering experiment.
