Advertisements
Advertisements
प्रश्न
Why is a cathode ray tube evacuated to a low pressure?
Advertisements
उत्तर
The cathode ray tube is evacuated to a low pressure to avoid collisions of electrons with air molecules.
APPEARS IN
संबंधित प्रश्न
State the energy conversion taking place in a cathode ray tube
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
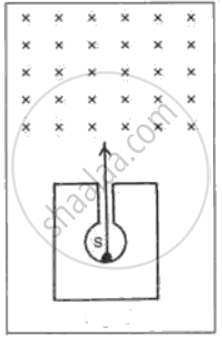
A certain nucleus A (mass number 238 and atomic number 92) is radioactive and becomes a nucleus B (mass number 234 and atomic number 90) by the loss of one particle.
What particle was emitted?
What is meant by Radioactivity?
Which part of the atom undergoes a change in the process of radioactive decay?
What is the value of the speed of gamma radiations in air or vacuum?
The half-life of a radioactive nuclide is 100 hours. The fraction of original activity that will remain after 150 hours would be ______.
A radioactive element is placed in an evacuated chamber. Then the rate of radioactive decay will ______.
Assertion (A): The force of repulsion between atomic nucleus and α-particle varies with distance according to inverse square law.
Reason (R): Rutherford did α-particles scattering experiment.
