Advertisements
Advertisements
प्रश्न
What is nucleus of an atom? Compare its size with that of the atom. Name its constitutents. How is the number of these constituents determined by the atomic number its atomic model.
Advertisements
उत्तर
The nucleus at the centre of atom, whose size is of the order of 10-15 m to 10-14 m.
The size of a nucleus is 10-5 to 10-4 times the size of an atom. It consists of protons and neutrons.
If Z is the atomic number and A is the mass number of an atom, then the atom contains Z number of electrons; Z number of protons and A - Z number of neutrons.
The atom is specified by the symbol \[\ce{^A_Z X}\] where X is the chemical symbol for the element.
संबंधित प्रश्न
Answer the following questions based on a hot cathode ray tube.
What will happen to the beam when it passes through the electric field?
What are free electrons?
Give a scientific explanation.
When the radiation coming out from a certain radioactive substance is passed through an electric field, marks are found at three places on the photographic plate placed in its path.
Give the relative ionizing power of α, β and γ radiations.
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
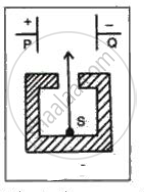
Why is the source S placed in a thick lead container?
What happens to the position of an element in the periodic table when it emits an alpha particle
Why should a radioactive substance not be touched by hands?
Justify with reason, whether the following nuclear reactions are allowed or not.
\[\ce{^A_Z X -> ^A_{Z - 2}X + ^4_{2}He}\]
Which part of the atom undergoes a change in the process of radioactive decay?
In U238 ore containing Uranium the ratio of U234 to Pb206 nuclei is 3. Assuming that all the lead present in the ore is final stable product of U238. Half life of U238 to be 4.5 × 109 years and the age of ore is ______ × 109 years. (in 109 years)
