Advertisements
Advertisements
Question
Define bound electrons.
Advertisements
Solution
As nucleus is positively charged it strongly attracts the negative charged electrons. The electron orbit close to the nucleus are tightly bound by strong attractive force of nucleus. These electrons are known as bound electrons.
RELATED QUESTIONS
What is thermionic emission?
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
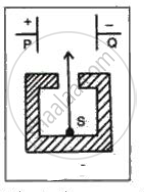
Why is the source S placed in a thick lead container?
What are the uses of radioactive isotopes in following?
(a) Medical field,
(b) Agriculture,
(c) Industries?
Define free electrons.
Why are materials of low work function preferred as thermionic cathode materials?
Which part of the atom undergoes a change in the process of radioactive decay?
From α, β and γ-rays, name the one which is not affected by electric or magnetic field?
Heavy radioactive elements eventually turn into ______.
