Advertisements
Advertisements
Question
In a cathode ray tube state the purpose of the fluorescent screen.
Advertisements
Solution
Fluorescent screen helps to obtain images on the screen by converting electrical signals to light (or) visual signals by ionizing radiations to light waves.
RELATED QUESTIONS
In a cathode ray tube, what is the function of anode?
An electromagnetic radiation is used for photography in fog. [2]
(i) Identify the radiation.
(ii) Why is this radiation mentioned by you, ideal for this purpose ?
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
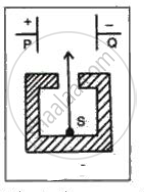
Why is the source S placed in a thick lead container?
A nucleus of an element X which has the symbol `""_84^202` X emits an alpha particle and then a beta particle. The final nucleus is `""_"b"^"a"` Y Find a and b.
State any two uses of cathode rays.
Justify with reason, whether the following nuclear reactions are allowed or not.
\[\ce{^A_Z X -> ^A_{Z - 2}X + ^4_{2}He}\]
Which part of the atom undergoes a change in the process of radioactive decay?
Mention one important function of ear main parts:
State the principle on which the functioning of a nuclear reactor is based.
The activity of a certain radionuclide decreases to 15 percent of its original value in 10 days. What is its half life?
[ln (0.15) = -1.9]
