Advertisements
Advertisements
Question
For a complex reaction:
(i) order of overall reaction is same as molecularity of the slowest step.
(ii) order of overall reaction is less than the molecularity of the slowest step.
(iii) order of overall reaction is greater than molecularity of the slowest step.
(iv) molecularity of the slowest step is never zero or non interger.
Advertisements
Solution
(i) order of overall reaction is same as molecularity of the slowest step.
(iv) molecularity of the slowest step is never zero or non interger.
Explanation:
(i) For a complex reaction, order of overall reaction = molecularity of slowest step. As rate of overall reaction depends upon total number of molecules involved in slowest step of the reaction. Hence, molecularity of the slowest step is equal to order of overall reaction.
(ii) Since the completion of any chemical reaction is not possible in the absence of reactants. Hence, slowest step of any chemical reaction must contain at least one reactant. Thus, molecularity’of the slowest step is never zero or non-integer.
APPEARS IN
RELATED QUESTIONS
In a first order reaction x → y, 40% of the given sample of compound remains unreacted in 45 minutes. Calculate rate constant of the reaction.
A → B is a first order reaction with rate 6.6 × 10-5m-s-1. When [A] is 0.6m, rate constant of the reaction is
- 1.1 × 10-5s-1
- 1.1 × 10-4s-1
- 9 × 10-5s-1
- 9 × 10-4s-1
Write molecularity of the following reaction:
2NO(g)+O2(g)→2NO2(g)
For a reaction A + B ⟶ P, the rate is given by
Rate = k [A] [B]2
How is the rate of reaction affected if the concentration of B is doubled?
For a chemical reaction R → P, the variation in the concentration (R) vs. time (t) plot is given as:

(i) Predict the order of the reaction.
(ii) What is the slope of the curve?
(iii) Write the unit of rate constant for this reaction.
For a reaction, \[\ce{A + B -> Product}\]; the rate law is given by, `r = k[A]^(1/2)[B]^2`. What is the order of the reaction?
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{CH3CHO_{(g)} -> CH4_{(g)} + CO_{(g)}}\] Rate = k [CH3CHO]3/2
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{C2H5Cl_{(g)} -> C2H4_{(g)} + HCl_{(g)}}\] Rate = k [C2H5Cl]
For the reaction:
\[\ce{2A + B → A2B}\]
the rate = k[A][B]2 with k = 2.0 × 10−6 mol−2 L2 s−1. Calculate the initial rate of the reaction when [A] = 0.1 mol L−1, [B] = 0.2 mol L−1. Calculate the rate of reaction after [A] is reduced to 0.06 mol L−1.
Mention the factors that affect the rate of a chemical reaction.
What is the order of a reaction which has a rate expression; Rate = `"k"["A"]^(3/2)["B"]^1`?
Rate law for the reaction \[\ce{A + 2B -> C}\] is found to be Rate = k [A][B]. Concentration of reactant ‘B’ is doubled, keeping the concentration of ‘A’ constant, the value of rate constant will be ______.
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?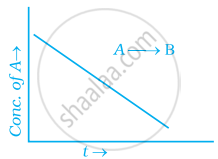
Assertion: Order and molecularity are same.
Reason: Order is determined experimentally and molecularity is the sum of the stoichiometric coefficient of rate determining elementary step.
A catalyst in a reaction changes which of the following?
The role of a catalyst is to change
In the presence of a catalyst, the heat evolved or absorbed during the reaction.
For a reaction R → p the concentration of reactant change from 0.03 m to 0.02 m in minute, calculate the average rate of the reaction using the unit of second.
If the 0.05 molar solution of m+ is replaced by a 0.0025 molar m+ solution, then the magnitude of the cell potential would be
Identify the order of reaction from the following unit for its rate constant:
L mol–1 s–1
