Advertisements
Advertisements
Question
For a reaction, \[\ce{A + B -> Product}\]; the rate law is given by, `r = k[A]^(1/2)[B]^2`. What is the order of the reaction?
Advertisements
Solution
The order of the reaction = `1/2+2`
= `2 1/2`
= 2.5
APPEARS IN
RELATED QUESTIONS
Write molecularity of the following reaction:
2NO(g)+O2(g)→2NO2(g)
For the first order thermal decomposition reaction, the following data were obtained:

Time / sec Totalpressure / atm
0 0.30
300 0.50
Calculate the rate constant
(Given: log 2 = 0.301, log3 = 0.4771, log 4 = 0.6021)
For the hydrolysis of methyl acetate in aqueous solution, the following results were obtained :
| t/s | 0 | 30 | 60 |
| [CH3COOCH3] / mol L–1 | 0.60 | 0.30 | 0.15 |
(i) Show that it follows pseudo first order reaction, as the concentration of water remains constant.
(ii) Calculate the average rate of reaction between the time interval 30 to 60 seconds.
(Given log 2 = 0.3010, log 4 = 0.6021)
The conversion of molecules X to Y follows second order kinetics. If concentration of X is increased to three times how will it affect the rate of formation of Y?
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{C2H5Cl_{(g)} -> C2H4_{(g)} + HCl_{(g)}}\] Rate = k [C2H5Cl]
In a reaction between A and B, the initial rate of reaction (r0) was measured for different initial concentrations of A and B as given below:
| A/mol L−1 | 0.20 | 0.20 | 0.40 |
| B/mol L−1 | 0.30 | 0.10 | 0.05 |
| r0/mol L−1 s−1 | 5.07 × 10−5 | 5.07 × 10−5 | 1.43 × 10−4 |
What is the order of the reaction with respect to A and B?
For a reaction R ---> P, half-life (t1/2) is observed to be independent of the initial concentration of reactants. What is the order of reaction?
Write the principle behind the following methods of refining:
Hydraulic washing
Molecularity of a reaction _____________.
Consider the reaction A ⇌ B. The concentration of both the reactants and the products varies exponentially with time. Which of the following figures correctly describes the change in concentration of reactants and products with time?
In any unimolecular reaction:
(i) only one reacting species is involved in the rate determining step.
(ii) the order and the molecularity of slowest step are equal to one.
(iii) the molecularity of the reaction is one and order is zero.
(iv) both molecularity and order of the reaction are one.
For which type of reactions, order and molecularity have the same value?
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?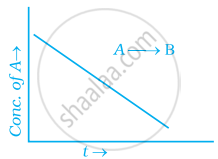
Assertion: Order of the reaction can be zero or fractional.
Reason: We cannot determine order from balanced chemical equation.
Assertion: The enthalpy of reaction remains constant in the presence of a catalyst.
Reason: A catalyst participating in the reaction, forms different activated complex and lowers down the activation energy but the difference in energy of reactant and product remains the same.
Use Molecular Orbital theory to determine the bond order in each of species, [He2j+ and [He2]2+?
The rate of a chemical reaction double for every 10° rise in temperature. If the temperature is raised. by 50°C, the rate of relation by about:-
For a reaction 1/2 A ⇒ 2B, rate of disappearance of A is related 't o the appearance of B by the expression:
For reaction 2A + B → BC + D which of the following does not Express the reaction rates
For a chemical reaction starting with some initial concentration of reactant At as a function of time (t) is given by the equation,
`1/("A"_"t"^4) = 2 + 1.5 xx 10^-3` t
The rate of disappearance of [A] is ____ × 10-2 M/sec when [A] = 2 M.
[Given: [At] in M and t in sec.]
[Express your answer in terms of 10-2 M /s]
[Round off your answer if required]
