Advertisements
Advertisements
Question
Explain the following:
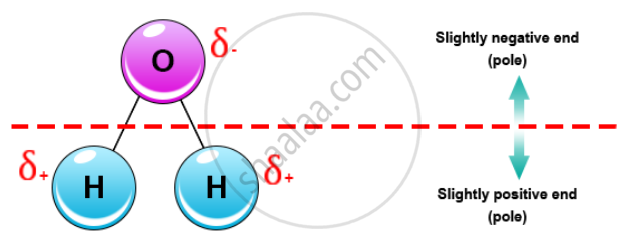
Water is a polar covalent molecule.
Advertisements
Solution
This means that in the water molecule, one side is positively charged and the other side is negatively charged. Since the water molecule (comprising two atoms of Hydrogen and one of Oxygen) is formed by covalent bonds, the electrons are shared.
This sharing causes the electrons to stay closer to the oxygen atom giving it a slight negative charge while the hydrogen atoms, as a consequence, have a slight positive charge.
APPEARS IN
RELATED QUESTIONS
What is the atomic number of carbon. Write its electronic configuration.
What type of bond exist in CCl4?
Explain why, ionic compounds conduct electricity in solution whereas covalent compounds do not conduct electricity.
Draw the electron-dot structure of H2O compound and state the type of bonding.
Explain why, diamond can be used in rock drilling equipment but graphite cannot.
The solution of one of the following compounds will not conduct electricity. This compounds is:
(a) NaCl
(b) CCl4
(c) MgCl2
(d) CaCl2
One of the following contains a double bond as well as single bonds. This is:
(a) CO2
(b) O2
(c) C2H4
(d) C2H2
State the type of bonding in the following molecule.
Hydroxyl ion
Explain the following briefly:
Sodium chloride dissolves in water but carbon tetra chloride is insoluble in water.
The electronic configuration of N2 is 2, 5. How many electrons in the outer shell of a N atom are not involved in the formation of a nitrogen molecule?
