Advertisements
Advertisements
Question
Explain the following:
Polar covalent compounds conduct electricity?
Advertisements
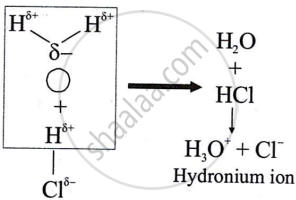
Solution
Polar covalent compounds conduct electricity because they form ions in their solutions and hence dissociate in water.
RELATED QUESTIONS
Why covalent compounds are different from ionic compounds?
Butanone is a four-carbon compound with the functional group:
What do you call the compounds having the same molecular formula but different structural arrangements of atoms?
What type of bonds are present in hydrogen chloride and oxygen?
State one test by which sodium chloride can be distinguished from sugar.
What do you understand by polar covalent compounds?
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Ethene
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Water
What is the term defined below?
A bond formed by a shared pair of electrons, each bonding atom contributing one electron to the pair.
Name the anion present in the following compound:
When a barium chloride solution is added to a solution of compound B, a white precipitate insoluble in dilute hydrochloric acid is formed.
