Advertisements
Advertisements
प्रश्न
Explain the following:
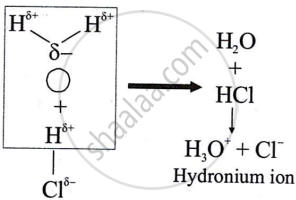
Polar covalent compounds conduct electricity?
Advertisements
उत्तर
Polar covalent compounds conduct electricity because they form ions in their solutions and hence dissociate in water.
संबंधित प्रश्न
Explain the nature of the covalent bond using the bond formation in CH3Cl.
Fill in the blank in the following sentence:
Two chlorine atoms combine to form a molecule. The bond between them is known as ..........
What type of bonding would you expect between Carbon and Chlorine?
State any two uses of graphite.
Give two example in following case:
Liquid non polar compounds
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Methanol
Choose the correct answer from the options given below
Which one is not example of polar covalent compound?
Potassium chloride is an electrovalent compound, while hydrogen chloride is a covalent compound, But, both conduct electricity in their aqueous solutions. Explain.
Write an Explanation.
Alkene
Carbon forms four covalent bonds by sharing its four valence electrons with four univalent atoms, e.g. hydrogen. After the formation of four bonds, carbon attains the electronic configuration of
