Advertisements
Advertisements
प्रश्न
Explain the following:
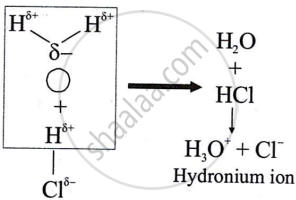
Polar covalent compounds conduct electricity?
Advertisements
उत्तर
Polar covalent compounds conduct electricity because they form ions in their solutions and hence dissociate in water.
APPEARS IN
संबंधित प्रश्न
State the reason to explain why covalent compounds "have low melting and boiling points."
Explain the bonding in methane molecule using electron dot structure.
What type of bonds are present in water molecule? Draw the electron-dot structure of water (H2O).
Give the formula of the compound that would be formed by the combination of the following pair of elements:
K and H
What is the difference between ionic compounds and polar covalent compounds?
Explain the following term with example.
Covalent bond
Taking hydrogen chloride and methane as examples, distinguish between a polar covalent bond and a non polar covalent bond.
Draw the electron dot diagram and structure of magnesium chloride.
State the type of bond formed, and draw Lewis structure of water.
Acetic acid was added to a solid X kept in a test tube. A colourless, odourless gas Y was evolved. The gas was passed through lime water, which turned milky. It was concluded that ______.
