Advertisements
Advertisements
प्रश्न
Explain the following:
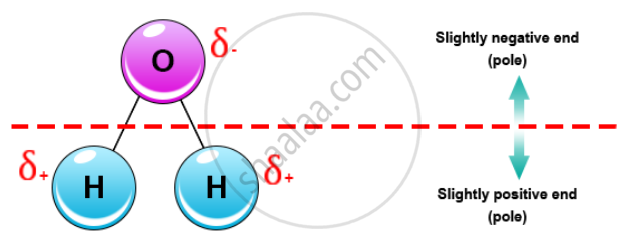
Water is a polar covalent molecule.
Advertisements
उत्तर
This means that in the water molecule, one side is positively charged and the other side is negatively charged. Since the water molecule (comprising two atoms of Hydrogen and one of Oxygen) is formed by covalent bonds, the electrons are shared.
This sharing causes the electrons to stay closer to the oxygen atom giving it a slight negative charge while the hydrogen atoms, as a consequence, have a slight positive charge.
APPEARS IN
संबंधित प्रश्न
Choose the correct answer from the options given below:
Which of the following is a common characteristic of a covalent compound?
1) high melting point
2) consists of molecules
3) always soluble in water
4) Conducts electricity when it is in the molten state
What type of bonds are present in water molecule? Draw the electron-dot structure of water (H2O).
Fill in the following blank with suitable word:
The form of carbon which is known as black lead is ...........
What type of bonding would you expect between Hydrogen and Chlorine?
Describe the structure of diamond. Draw a simple diagram to show the arrangement of carbon atoms in diamond.
Explain the following:
Covalent compounds are generally gases or liquids or soft solids.
Name two compounds that are covalent when taken pure but produce ions when dissolved in water.
List two differences between the properties exhibited by covalent compounds and ionic compounds.
It is observed that covalent compounds are had conductors of electricity. Give reason.
