Advertisements
Advertisements
प्रश्न
Explain the following:
Water is a polar covalent molecule.
Advertisements
उत्तर
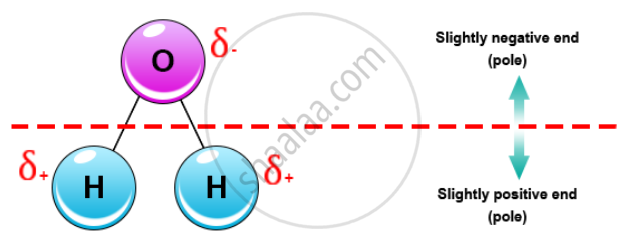
This means that in the water molecule, one side is positively charged and the other side is negatively charged. Since the water molecule (comprising two atoms of Hydrogen and one of Oxygen) is formed by covalent bonds, the electrons are shared.
This sharing causes the electrons to stay closer to the oxygen atom giving it a slight negative charge while the hydrogen atoms, as a consequence, have a slight positive charge.
APPEARS IN
संबंधित प्रश्न
What would be the electron dot structure of carbon dioxide which has the formula CO2?
Give one example of a molecule containing a triple covalent bond.
Fill in the blank in the following sentence:
Two atoms of the same element combine to form a molecule. The bond between them is known as ................ bond.
Describe the structure of diamond. Draw a simple diagram to show the arrangement of carbon atoms in diamond.
One of the following contains a double bond as well as single bonds. This is:
(a) CO2
(b) O2
(c) C2H4
(d) C2H2
Give the formulae of the chlorides of the elements X and Y having atomic numbers of 3 and 6 respectively. Will the properties of the two chlorides be similar or different? Explain your answer.
Draw all possile structural formulae of compound from their molecular formula given below.
C4H10
(a) Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the choices (a), (b), (c) and (d) given below
In the liquid state, X will
From the following hydrocarbon _______ is the cyclic hydrocarbon.
Which of the following is the purest form of carbon?
