Advertisements
Advertisements
प्रश्न
Give one example of a molecule containing a triple covalent bond.
Advertisements
उत्तर
A molecule containing a triple covalent bond: nitrogen molecule 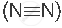 )
)
APPEARS IN
संबंधित प्रश्न
What are covalent compounds?
Elements forming ionic compounds attain noble gas configuration by either gaining or losing electrons from their outermost shells. Give reason to explain why carbon cannot attain noble gas configuration in this manner to form its compounds.
Choose the correct answer from the options given below:
Which of the following is a common characteristic of a covalent compound?
1) high melting point
2) consists of molecules
3) always soluble in water
4) Conducts electricity when it is in the molten state
Buckminsterfullerene is spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many pentagons of carbon atoms are present in one molecule of buckminsterfullerene?
What is a covalent bond?
Give two general properties of ionic compounds and two those of covalent compounds.
Describe the structure of graphite with the help of a labelled diagram.
The solution of one of the following compounds will not conduct electricity. This compounds is:
(a) NaCl
(b) CCl4
(c) MgCl2
(d) CaCl2
Buckminsterfullerene is an allotropic form of the element:
(a) phoshorus
(b) fluorine
(c) carbon
(d) sulphur
Give two example in following case:
Solid covalent compounds
Give two example in following case:
Gaseous non polar compounds
Explain the following briefly:
Sodium chloride dissolves in water but carbon tetra chloride is insoluble in water.
Choose the correct answer from the options given below
Condition favorable for formation of a covalent bond is
Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the options A, B, C and D given below:
X is likely to have a ______.
Write an Explanation.
Alkene
List two differences between the properties exhibited by covalent compounds and ionic compounds.
Acetic acid was added to a solid X kept in a test tube. A colourless, odourless gas Y was evolved. The gas was passed through lime water, which turned milky. It was concluded that ______.
Mineral acids are stronger acids than carboxylic acids because
- mineral acids are completely ionised
- carboxylic acids are completely ionised
- mineral acids are partially ionised
- carboxylic acids are partially ionised
"Carbon prefers to share its valence electrons with other atoms of carbon or with atoms of other elements rather than gaining or losing the valence electrons in order to attain noble gas configuration." Give reasons to justify this statement.
The electron dot structure of chlorine molecule is:
