Advertisements
Advertisements
प्रश्न
Give one example of a molecule containing a double covalent bond
Advertisements
उत्तर
A molecule containing a double covalent bond: oxygen molecule 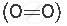
APPEARS IN
संबंधित प्रश्न
State the reason why carbon can neither form C4+ cations nor C4− anions but forms covalent compounds.
What type of bond is present in oxygen molecule?
Why does carbon form compounds mainly by covalent bonding?
Give one example, state what are covalent compounds?
How buckminsterfullerene is it related to diamond and graphite?
Give two general properties of ionic compounds and two those of covalent compounds.
Give the formula of the compound that would be formed by the combination of the following pair of elements:
Al and Cl2
Write two points of difference in the structures of diamond and graphite.
Out of KCl, HCl, CCl4 and NaCl, the compounds which are not ionic are:
(a) KCl and HCl
(b) HCl and CCl4
(c) CCl4 and NaCl
(d) KCl and CCl4
A hydrocarbon having one double bond has 100 carbon atoms in its molecule. The number of hydrogen atoms in its molecule will be
(a) 200
(b) 198
(c) 202
(d) 196
Explain the Structural isomerism term with example.
Explain the following term with example.
Covalent bond
Explain the following term with example.
Alkane
Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the options A, B, C and D given below:
The type of bonding in X will be ______.
Name a greenish-yellow gas which also bleaches.
From the following hydrocarbon _______ is the cyclic hydrocarbon.
Mineral acids are stronger acids than carboxylic acids because
- mineral acids are completely ionised
- carboxylic acids are completely ionised
- mineral acids are partially ionised
- carboxylic acids are partially ionised
Molecular reactions are ______ in the covalent compound.
"Carbon prefers to share its valence electrons with other atoms of carbon or with atoms of other elements rather than gaining or losing the valence electrons in order to attain noble gas configuration." Give reasons to justify this statement.
Non-polar covalent compounds are ______ conductors of heat and electricity.
