Advertisements
Advertisements
Question
Give one example of a molecule containing a double covalent bond
Advertisements
Solution
A molecule containing a double covalent bond: oxygen molecule 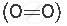
APPEARS IN
RELATED QUESTIONS
Name the element whose one of the allotropic forms is buckminsterfullerene.
State one test by which sodium chloride can be distinguished from sugar.
Which inert gas does the Cl atom in HCl resemble in electron arrangement?
What is the difference between ionic compounds and covalent compounds?
Elements forming ionic compounds attain noble gas electronic configuration by either gaining or losing electrons from their valence shells. Explain giving reason why carbon cannot attain such a configuration in this manner to form its compounds. Name the type of bonds formed in ionic compounds and in the compounds formed by carbon. Also explain the reason why carbon compounds are generally poor conductors of electricity.
Distinguish between ionic and covalent compounds under the following properties:
(i) Strength of forces between constituent elements
(ii) Solubility of compounds in water
(iii) Electrical conduction in substances
Explain the Structural isomerism term with example.
Choose and write the correct option.
Which type of carbon-carbon bonds are present in Vanaspati ghee?
Explain the following:
Covalent compounds have low melting and boiling point.
What is the term defined below?
A bond formed by a shared pair of electrons, each bonding atom contributing one electron to the pair.
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2,8,1 |
2,8,7 |
2,5 |
1 |
Answer the following questions based on the table above:
What type of bond is formed between Y and Z.
Draw the electron dot diagram and structure of magnesium chloride.
Explain the following:
Water is a polar covalent molecule.
Name the anion present in the following compound:
When a barium chloride solution is added to a solution of compound B, a white precipitate insoluble in dilute hydrochloric acid is formed.
Name the types of Hydrocarbons.
Generally, the melting and boiling point of carbon compounds are found to be less than _______ °C.
From the following hydrocarbon _______ is the cyclic hydrocarbon.
Write the molecular formula of the given compound.
Propylene
Give an example for each of the following statement.
Formation of coordinate covalent bond between NH3 ➝ BF3 molecules
Although metals form basic oxides, which of the following metals form an amphoteric oxide?
