Advertisements
Advertisements
Question
Give one example of a molecule containing a triple covalent bond.
Advertisements
Solution
A molecule containing a triple covalent bond: nitrogen molecule 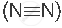 )
)
APPEARS IN
RELATED QUESTIONS
Why covalent compounds are different from ionic compounds?
Explain the bonding in methane molecule using electron dot structure.
Name the black substance of pencil. Will the current flow through the electrical circuit when we use the sharpened ends of the pencil to complete the circuit?
What type of bonds are present in methane (CH4) and sodium chloride (NaCl)?
Draw the electron-dot structure of HCl compound and state the type of bonding.
Why is graphite a good conductor of electricity but diamond is a non-conductor of electricity?
One of the following contains a double bond as well as single bonds. This is:
(a) CO2
(b) O2
(c) C2H4
(d) C2H2
The electronic configurations of two elements A and B are given below:
| A | 2, 6 |
| B | 2, 8, 1 |
(a) What type of chemical bond is formed between the two atoms of A?
(b) What type of chemical bond will be formed between the atoms of A and B?
Give two example in following case:
Gaseous polar compounds
Explain the following term with example.
Alkane
Explain the following term with example.
Unsaturated hydrocarbon
Observe the straight chain hydrocarbons given below and answer the following
questions :
i) Which of the straight chain compounds from A and B is saturated and unsaturated straight chains?
ii) Name these straight chains.
iii) Write their chemical formulae and number of – CH2 - units.

Explain the following briefly.
Pure water does not conduct electricity, but on adding sodium chloride to it, it starts conducting electricity.
(a) Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the choices (a), (b), (c) and (d) given below
The type of bonding in X will be
Give an example of the covalent bond formed by
(i) Similar atoms (ii) Dissimilar atoms
In the formation of electrovalent compounds, electrons are transferred from one element to another. How are electrons involved in the formation of a covalent compound?
Explain covalent bond with example.
Statement (A): Covalent compounds are bad conductors of electricity.
Reason (B): Covalent compounds contain charged particles (ions)
Show the covalent bond formation in nitrogen molecule.
