Advertisements
Advertisements
प्रश्न
Give one example of a molecule containing a triple covalent bond.
Advertisements
उत्तर
A molecule containing a triple covalent bond: nitrogen molecule 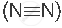 )
)
APPEARS IN
संबंधित प्रश्न
Elements forming ionic compounds attain noble gas configuration by either gaining or losing electrons from their outermost shells. Give reason to explain why carbon cannot attain noble gas configuration in this manner to form its compounds.
Name the type of bonds formed in ionic compounds and in the compounds formed by carbon.
Why are most carbon compounds poor conductors of electricity?
Buckminsterfullerene is spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many hexagons of carbon atoms are present in one molecule of buckminsterfullerene?
What type of bonds are present in hydrogen chloride and oxygen?
Describe the structure of graphite with the help of a labelled diagram.
State any two uses of graphite.
A hydrocarbon having one double bond has 100 carbon atoms in its molecule. The number of hydrogen atoms in its molecule will be
(a) 200
(b) 198
(c) 202
(d) 196
Elements forming ionic compounds attain noble gas electronic configuration by either gaining or losing electrons from their valence shells. Explain giving reason why carbon cannot attain such a configuration in this manner to form its compounds. Name the type of bonds formed in ionic compounds and in the compounds formed by carbon. Also explain the reason why carbon compounds are generally poor conductors of electricity.
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Methanol
The covalent bond in which the electrons are shared equally between the combining atoms.
Explain the following:
Non-polar covalent compounds are insoluble in water.
Element A has 2 electrons in its M shell. Element B has atomic number 7.
Write equations to show how A and B form ions.
Name a greenish-yellow gas which also bleaches.
Fill in the blank and rewrite the completed statement:
Covalent compounds are generally soluble in _________ solvents.
Name the types of Hydrocarbons.
The molecular masses of a carbon compound spread over a range of _______.
Write a short note.
Aromatic hydrocarbons
`"CH"_3 - "CH"_2 - "OH" overset("Alkaline""KMnO"_4 + "Heat")(->) "CH"_3 - "COOH"`
In the above given reaction, alkaline KMnO4 acts as
What is a covalent bond?
