Advertisements
Advertisements
प्रश्न
Give one example of a molecule containing a double covalent bond
Advertisements
उत्तर
A molecule containing a double covalent bond: oxygen molecule 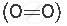
APPEARS IN
संबंधित प्रश्न
State whether the following statement is true or false:
Diamond and graphite are the covalent compounds of carbon element (C)
Name the hardest natural substance known.
What type of chemical bond is formed between carbon and bromine?
What type of bonds are present in water molecule? Draw the electron-dot structure of water (H2O).
What type of bonds are present in H2 molecule? Draw their electron-dot structures.
What is buckminsterfullerene?
Name one covalent compound containing chlorine.
Explain why, diamond is hard while graphite is soft (though both are made of carbon atoms).
State one use of diamond which depends on its 'extraordinary brilliance' and one use of graphite which depends on its being 'black and quite soft'.
Define a covalent bond.
Complete the following:
When the nuclei of two different reacting atoms are of ______ mass, then a bond so formed is called ______ covalent bond. (equal, unequal, polar, non-polar)
What do you understand by lone pair and shared pair?
State the type of bond formed when the combining atom has small E.N. difference.
Name two compounds that are covalent when taken pure but produce ions when dissolved in water.
From the following hydrocarbon _______ is the cyclic hydrocarbon.
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
| \[\begin{array}{cc} \phantom{.........}\ce{H}\\ \phantom{.........}|\\ \ce{H - C ≡ C - C - H}\\ \phantom{.........}|\\ \phantom{.........}\ce{H} \end{array}\] |
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
Carbon forms four covalent bonds by sharing its four valence electrons with four univalent atoms, e.g., hydrogen. After the formation of four bonds, carbon attains the electronic configuration of ______.
Which of the following are correct structural isomers of butane?
- \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - H}\\
|\phantom{.....}|\phantom{.....}|\\
\ce{H}\ce{H-C-H}\ce{H}\\
|\\
\ce{H}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - H}\\
|\phantom{.....}\backslash\phantom{..}|\\
\phantom{....}\ce{H}\phantom{......}\ce{C - H}\phantom{}\\
\phantom{.......}|\\
\phantom{.......}\ce{H}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\\
\ce{H - C - C - H}\\
|\phantom{....}|\\
\ce{H - C - C - H}\\
|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\\
\end{array}\]
