Advertisements
Advertisements
Question
[Cr(NH3)6]3+ is paramagnetic while [Ni(CN)4]2− is diamagnetic. Explain why?
Advertisements
Solution
Formation of [Cr(NH3)6]3+: The oxidation state of chromium in [Cr(NH3)6]3+ ion is +3. The electronic configuration of chromium is [Ar] 3d54s1. The hybridisation is shown in the following diagram:

The Cr3+ ion provides six vacant orbitals to accommodate six electron pairs from six ammonia molecules. As a result, the complex [Cr(NH3)6]3+ has d2sp3 hybridisation and is octahedral. The presence of three unpaired electrons in the complex explains its paramagnetic property.
Formation of [Ni(CN)4]2−: In [Ni(CN)4]2− the oxidation state of Ni is +2 and its electronic configuration is 3d. Hybridisation can be explained as follows:
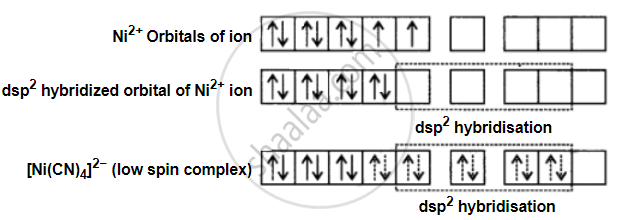
Each hybridised orbital accepts a pair of electrons from the cyanide ion. The absence of unpaired electrons confirms the diamagnetic behaviour of [Ni(CN)4]2−.
APPEARS IN
RELATED QUESTIONS
Predict the number of unpaired electrons in the square planar [Pt(CN)4]2− ion.
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[Fe(CN)6]4−
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[Co(C2O4)3]3−
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[CoF6]3−
Write the hybridisation and number of unpaired electrons in the complex `[CoF_6]^(3-)`. (Atomic No. of Co = 27)
[NiCl4]2- is paramagnetic while [Ni(CO)4] is diamagnetic though both are tetrahedral. Why? (Atomic no. Ni = 28)
Using valence bond theory, explain the following in relation to the complexes given below:
\[\ce{[Co(NH3)6]^{3+}}\]
(i) Type of hybridisation.
(ii) Inner or outer orbital complex.
(iii) Magnetic behaviour.
(iv) Spin only magnetic moment value.
In a coordination entity, the electronic configuration of the central metal ion is t2g3 eg1
Is the coordination compound a high spin or low spin complex?
How many radial nodes for 3p orbital?
Which of the statement given below is incorrect about H2O2?
As the s-character of hybridised orbital increases, the bond angle
In Fe(CO)5, the Fe – C bond possesses
Using valence bond theory, predict the hybridization and magnetic character of the following:
[CoF6]3– [Atomic number of Co = 27]
The magnetic moment of [NiCl4]2− is ______.
[Atomic number: Ni = 28]
[Ni(CO)4] has tetrahedral geometry while [Ni(CN)4]2− has square planar, yet both exhibit diamagnetism. Explain.
[Atomic number: Ni = 28]
The geometry and magnetic behaviour of the complex [Ni(CO)4] are ______.
Given below are two statements:
Statement I: Both [Co(NH3)6]3+ and [CoF6]3− complexes are octahedral but differ in their magnetic behavior.
Statement II: [Co(NH3)6]3+ is diamagnetic whereas [CoF6]3− is paramagnetic.
In the light of the above statements, Choose the correct answer form the options given below:
