Advertisements
Advertisements
प्रश्न
[Cr(NH3)6]3+ is paramagnetic while [Ni(CN)4]2− is diamagnetic. Explain why?
Advertisements
उत्तर
Formation of [Cr(NH3)6]3+: The oxidation state of chromium in [Cr(NH3)6]3+ ion is +3. The electronic configuration of chromium is [Ar] 3d54s1. The hybridisation is shown in the following diagram:

The Cr3+ ion provides six vacant orbitals to accommodate six electron pairs from six ammonia molecules. As a result, the complex [Cr(NH3)6]3+ has d2sp3 hybridisation and is octahedral. The presence of three unpaired electrons in the complex explains its paramagnetic property.
Formation of [Ni(CN)4]2−: In [Ni(CN)4]2− the oxidation state of Ni is +2 and its electronic configuration is 3d. Hybridisation can be explained as follows:
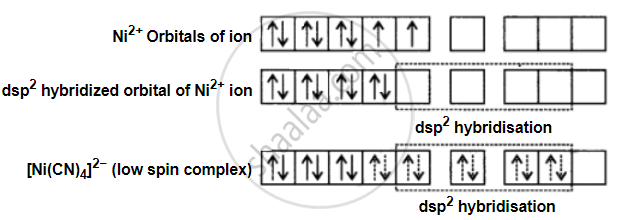
Each hybridised orbital accepts a pair of electrons from the cyanide ion. The absence of unpaired electrons confirms the diamagnetic behaviour of [Ni(CN)4]2−.
APPEARS IN
संबंधित प्रश्न
On the basis of valence bond theory explain the nature of bonding in [CoF6]3 ion.
[NiCl4]2− is paramagnetic, while [Ni(CO)4] is diamagnetic, though both are tetrahedral. Why? (Atomic number of Ni = 28)
Predict the number of unpaired electrons in the square planar [Pt(CN)4]2− ion.
Explain the geometry of `[Co(NH_3)_6]^(3+)` on the basis of hybridisation. (Z of Co = 27)
Write the hybridisation and number of unpaired electrons in the complex `[CoF_6]^(3-)`. (Atomic No. of Co = 27)
[NiCl4]2- is paramagnetic while [Ni(CO)4] is diamagnetic though both are tetrahedral. Why? (Atomic no. Ni = 28)
Using valence bond theory, explain the following in relation to the complexes given below:
\[\ce{[Cr(H2O)6]^{3+}}\]
(i) Type of hybridisation.
(ii) Inner or outer orbital complex.
(iii) Magnetic behaviour.
(iv) Spin only magnetic moment value.
Using valence bond theory, explain the following in relation to the complexes given below:
\[\ce{[FeCl6]^{4-}}\]
(i) Type of hybridisation.
(ii) Inner or outer orbital complex.
(iii) Magnetic behaviour.
(iv) Spin only magnetic moment value.
The type of hybridization involved in Octahedral complexes is ______.
Write the hybridization and shape of the following complexes:
[Ni(CN)4]2−
In a coordination entity, the electronic configuration of the central metal ion is t2g3 eg1
Is the coordination compound a high spin or low spin complex?
If orbital quantum number (l) has values 0, 1, 2 and 3, deduce the corresponding value of principal quantum number, n.
When the hybridization state of carbon changes from sp3 to sp2 and finally to sp, the angle between hybridized orbital will
What is the no. of possible isomers for the octahedral complex [Co(NH3)2(C2O4)2]?
In Fe(CO)5, the Fe – C bond possesses
Valence bond theory is based on the assumption that the bonds formed between the metal ions and ligands are ______
According to the valence bond theory, the hybridization of central metal atom is dsp2 for which one of the following compounds?
Given below are two statements:
Statement I: Both [Co(NH3)6]3+ and [CoF6]3− complexes are octahedral but differ in their magnetic behavior.
Statement II: [Co(NH3)6]3+ is diamagnetic whereas [CoF6]3− is paramagnetic.
In the light of the above statements, Choose the correct answer form the options given below:
