Advertisements
Advertisements
प्रश्न
[NiCl4]2- is paramagnetic while [Ni(CO)4] is diamagnetic though both are tetrahedral. Why? (Atomic no. Ni = 28)
Advertisements
उत्तर
[NiCl4]2-
Ni is in + 2 oxidation state.
3d8 configuration 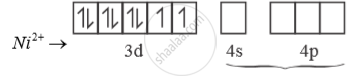
Cl- is weak field ligand. So, pairing doesnot occur.
sp3 hybridised orbitals of 2 Ni2+ 

[ ] NiCl is paramagnetic as n = 2
4 [ ( ) ] Ni CO Ni is in ‘0’ oxidation state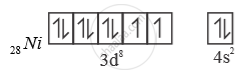
In presence of ‘CO’ pairing of e– takes place ‘CO’ is strong field ligand. So, with ‘CO
[Ni(CO)4] → 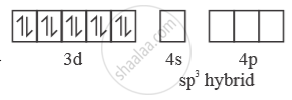
[Ni(CO)4] → 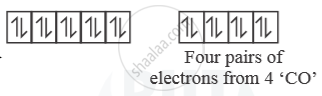
So, [Ni(CO)4] is diamagnetic as n = 0.
APPEARS IN
संबंधित प्रश्न
On the basis of valence bond theory explain the nature of bonding in [CoF6]3 ion.
Explain on the basis of valence bond theory that [Ni(CN)4]2− ion with square planar structure is diamagnetic and the [NiCl4]2− ion with tetrahedral geometry is paramagnetic.
Discuss the nature of bonding in the following coordination entity on the basis of valence bond theory:
[FeF6]3−
Write the hybridisation and number of unpaired electrons in the complex `[CoF_6]^(3-)`. (Atomic No. of Co = 27)
Using valence bond theory, explain the following in relation to the complexes given below:
\[\ce{[Cr(H2O)6]^{3+}}\]
(i) Type of hybridisation.
(ii) Inner or outer orbital complex.
(iii) Magnetic behaviour.
(iv) Spin only magnetic moment value.
Using valence bond theory, explain the following in relation to the complexes given below:
\[\ce{[FeCl6]^{4-}}\]
(i) Type of hybridisation.
(ii) Inner or outer orbital complex.
(iii) Magnetic behaviour.
(iv) Spin only magnetic moment value.
Which of the statement given below is incorrect about H2O2?
Which of the following has square planar structures?
[Ni(CO)4] has tetrahedral geometry while [Ni(CN)4]2− has square planar, yet both exhibit diamagnetism. Explain.
[Atomic number: Ni = 28]
Which of the following are paramagnetic?
- [NiCl4]2−
- Ni(CO)4
- [Ni(CN)4]2−
- [Ni(H2O)6]2+
- Ni(PPh3)4
Choose the correct answer from the options given below:
