Advertisements
Advertisements
प्रश्न
Write the electronic configuration of Fe(III) on the basis of crystal field theory when it forms an octahedral complex in the presence of (i) strong field, and (ii) weak field ligand. (Atomic no.of Fe=26)
Advertisements
उत्तर
`"Fe"= ["Ar"]3"d"^6 4"s"^2`
`"Fe"^{3+} = ["Ar"]3"d"^6 4"s"^2`
`"Fe"^(+3) = ["AR"] 3"d"^5 4"s"^0`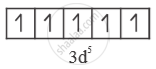
In octahedral crystal field.
(i) Strong field ligand - Electrons will be paired.
No. of unpaired e- = 1
Paramagnetic
`μ = sqrt(n(n+2) = sqrt(1(3) = sqrt3 B.M`
(ii) Weak field ligand
- Electrons follow Hund’s rule 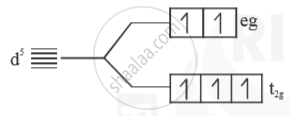
No. of unpaired = 5
Paramagnetic
`μ =sqrt(5(5+2))= sqrt(35) B.M.`
APPEARS IN
संबंधित प्रश्न
On the basis of crystal field theory, write the electronic configuration for d4 ion if ∆0 < P.
The hexaquo manganese(II) ion contains five unpaired electrons, while the hexacyanoion contains only one unpaired electron. Explain using Crystal Field Theory.
Complete and balance the following reactions:
\[\ce{P4 + H2SO4 ->}\] ______ + ______ + ______
Atomic number of \[\ce{Mn, Fe, Co}\] and Ni are 25, 26, 27 and 28 respectively. Which of the following outer orbital octahedral complexes have same number of unpaired electrons?
(i) \[\ce{[MnCl6]^{3-}}\]
(ii) \[\ce{[FeF6]^{3-}}\]
(iii) \[\ce{[CoF6]^{3-}}\]
(iv) \[\ce{[Ni(NH3)6]^{2+}}\]
The correct order of increasing crystal field strength in following series:
The CFSE of [CoCl6]3– is 18000 cm–1 the CFSE for [CoCl4]– will be ______.
The correct order of intensity of colors of the compounds is ______.
Consider that d6 metal ion (M2+) forms a complex with aqua ligands and the spin only magnetic moment of the complex is 4.90 BM. The geometry and the crystal field stabilization energy of the complex are ______.
On the basis of crystal field theory, write the electronic configuration for the d5 ion with a weak ligand for which Δ0 < P.
The value of the spin only magnetic moment for one of the following configurations is 2.84 BM. The correct one is:
