Advertisements
Advertisements
प्रश्न
Write the electronic configuration of Fe(III) on the basis of crystal field theory when it forms an octahedral complex in the presence of (i) strong field, and (ii) weak field ligand. (Atomic no.of Fe=26)
Advertisements
उत्तर
`"Fe"= ["Ar"]3"d"^6 4"s"^2`
`"Fe"^{3+} = ["Ar"]3"d"^6 4"s"^2`
`"Fe"^(+3) = ["AR"] 3"d"^5 4"s"^0`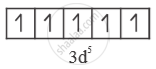
In octahedral crystal field.
(i) Strong field ligand - Electrons will be paired.
No. of unpaired e- = 1
Paramagnetic
`μ = sqrt(n(n+2) = sqrt(1(3) = sqrt3 B.M`
(ii) Weak field ligand
- Electrons follow Hund’s rule 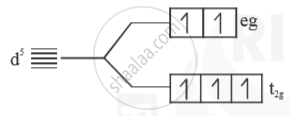
No. of unpaired = 5
Paramagnetic
`μ =sqrt(5(5+2))= sqrt(35) B.M.`
APPEARS IN
संबंधित प्रश्न
State the superiority of crystal field theory over valence bond theory.
Complete and balance the following reactions:
\[\ce{P4 + H2SO4 ->}\] ______ + ______ + ______
The CFSE for octahedral \[\ce{[CoCl6]^{4-}}\] is 18,000 cm–1. The CFSE for tetrahedral \[\ce{[CoCl4]^{2-}}\] will be ______.
Atomic number of \[\ce{Mn, Fe, Co}\] and Ni are 25, 26, 27 and 28 respectively. Which of the following outer orbital octahedral complexes have same number of unpaired electrons?
(i) \[\ce{[MnCl6]^{3-}}\]
(ii) \[\ce{[FeF6]^{3-}}\]
(iii) \[\ce{[CoF6]^{3-}}\]
(iv) \[\ce{[Ni(NH3)6]^{2+}}\]
An aqueous pink solution of cobalt (II) chloride changes to deep blue on addition of excess of HCl. This is because:
(i) \[\ce{[Co(H2O)6]^{2+}}\] is transformed into \[\ce{[CoCl6]}^{4-}\]
(ii) \[\ce{[Co(H2O)6]^{2+}}\] is transformed into \[\ce{[CoCl4]}^{2-}\]
(iii) tetrahedral complexes have smaller crystal field splitting than octahedral complexes.
(iv) tetrahedral complexes have larger crystal field splitting than octahedral complex.
\[\ce{CuSO4 . 5H2O}\] is blue in colour while \[\ce{CuSO4}\] is colourless. Why?
Why are different colours observed in octahedral and tetrahedral complexes for the same metal and same ligands?
Crystal field stabilising energy for high spind4 octahedral complex is:-
What is crystal field splitting energy?
The value of the spin only magnetic moment for one of the following configurations is 2.84 BM. The correct one is:
