Advertisements
Advertisements
Question
Consider a thin target (10–2 cm square, 10–3 m thickness) of sodium, which produces a photocurrent of 100 µA when a light of intensity 100W/m2 (λ = 660 nm) falls on it. Find the probability that a photoelectron is produced when a photons strikes a sodium atom. [Take density of Na = 0.97 kg/m3].
Advertisements
Solution
According to the problem, the area of the target A = 10–2 cm2 = 10–4 m2
And thickness, d = 10–3 m
Photocurrent, i = 100 × 10–6 A= 10–4 A
Intensity, I = 100 W/m2
⇒ λ = 660 nm = 660 × 10–9 m
ρNa = 0.97 kg/m3
Avogadro number = 6 × 1026 kg atom
Volume of sodium target = A × d
= 10–4 × 10–3
= 10–7 m3
We know that 6 × 1026 atoms of sodium weigh = 23 kg
Density of sodium = 0.97 kg/m3
Hence the volume of 6 × 1026 sodium atoms = `23/0.97` m3
Volume occupied by one sodium atom = `23/(0.97 xx (6 xx 10^36))` = 3.95 × 10–26 m3
Number of sodium atoms in target `(N_"sodium") = 10^-7/(3.95 xx 10^-26)` = 2.53 × 1018
Let m be the number of photons falling per second on the target.
Energy of each photon = `(hc)/A`
Total energy falling per second on target = `(nhc)/λ = IA`
∴ `n = (IAλ)/(hc)`
= `(100 xx 10^-4 xx (660 xx 10^-9))/((6.62 xx 10^-34) xx (3 xx 10^8))` = 3.3 × 1016
Let P be the probability of emission per atom per photon. The number of photoelectrons emitted per second
`N = P xx n xx (N_"sodium")`
= `P xx (33 xx 10^16) xx (2.53 xx 10^18)`
Now, according to the question,
i = 100 µA = 100 × 10–6 = 10–4 A
Current, i = Ne
∴ `10^-4 xx P xx (3.3 xx 10^16) xx (2.53 xx 10^18) xx (1.6 xx 10^-19)`
⇒ `P = 10^-4/((3.3 xx 10^16) xx (2.53 xx 10^18) xx (1.6 xx 10^-19))`
= 7.48 × 10–21
Then, the probability of photoemission by a single photon on a single atom is very much less than 1. Because the absorption of two photons by an atom is negligible.
APPEARS IN
RELATED QUESTIONS
The photoelectric cut-off voltage in a certain experiment is 1.5 V. What is the maximum kinetic energy of photoelectrons emitted?
Use the same formula you employ in (a) to obtain electron speed for an collector potential of 10 MV. Do you see what is wrong? In what way is the formula to be modified?
Ultraviolet light of wavelength 2271 Å from a 100 W mercury source irradiates a photo-cell made of molybdenum metal. If the stopping potential is −1.3 V, estimate the work function of the metal. How would the photo-cell respond to a high intensity (∼105 W m−2) red light of wavelength 6328 Å produced by a He-Ne laser?
A mercury lamp is a convenient source for studying frequency dependence of photoelectric emission, since it gives a number of spectral lines ranging from the UV to the red end of the visible spectrum. In our experiment with rubidium photo-cell, the following lines from a mercury source were used:
λ1 = 3650 Å, λ2 = 4047 Å, λ3 = 4358 Å, λ4 = 5461 Å, λ5 = 6907 Å,
The stopping voltages, respectively, were measured to be:
V01 = 1.28 V, V02 = 0.95 V, V03 = 0.74 V, V04 = 0.16 V, V05 = 0 V
Determine the value of Planck’s constant h, the threshold frequency and work function for the material.
[Note: You will notice that to get h from the data, you will need to know e (which you can take to be 1.6 × 10−19 C). Experiments of this kind on Na, Li, K, etc. were performed by Millikan, who, using his own value of e (from the oil-drop experiment) confirmed Einstein’s photoelectric equation and at the same time gave an independent estimate of the value of h.]
The threshold wavelength of a metal is λ0. Light of wavelength slightly less than λ0 is incident on an insulated plate made of this metal. It is found that photoelectrons are emitted for some time and after that the emission stops. Explain.
If an electron has a wavelength, does it also have a colour?
Two photons of
Light of wavelength λ falls on a metal with work-function hc/λ0. Photoelectric effect will take place only if
If the wavelength of light in an experiment on photoelectric effect is doubled,
(a) photoelectric emission will not take place
(b) photoelectric emission may or may not take place
(c) the stopping potential will increase
(d) the stopping potential will decrease
A 100 W light bulb is placed at the centre of a spherical chamber of radius 20 cm. Assume that 60% of the energy supplied to the bulb is converted into light and that the surface of the chamber is perfectly absorbing. Find the pressure exerted by the light on the surface of the chamber.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Find the maximum kinetic energy of the photoelectrons ejected when light of wavelength 350 nm is incident on a cesium surface. Work function of cesium = 1.9 eV
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Answer the following question.
Plot a graph of photocurrent versus anode potential for radiation of frequency ν and intensities I1 and I2 (I1 < I2).
Define the terms "stopping potential' and 'threshold frequency' in relation to the photoelectric effect. How does one determine these physical quantities using Einstein's equation?
Do all the electrons that absorb a photon come out as photoelectrons?
Consider a metal exposed to light of wavelength 600 nm. The maximum energy of the electron doubles when light of wavelength 400 nm is used. Find the work function in eV.
How would the stopping potential for a given photosensitive surface change if the frequency of the incident radiation were increased? Justify your answer.
The figure shows a plot of stopping potential (V0) versus `1/lambda`, where λ is the wavelength of the radiation causing photoelectric emission from a surface. The slope of the line is equal to ______.

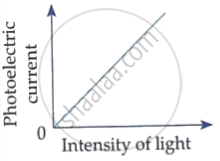
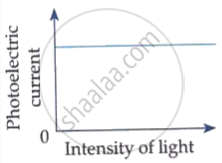
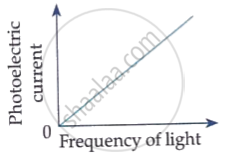
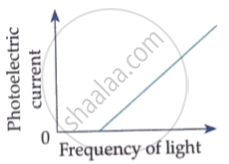
Which of the following options represents the variation of photoelectric current with property of light shown on the x-axis?