Advertisements
Advertisements
Question
A radio can tune in to any station in the 7.5 MHz to 12 MHz band. What is the corresponding wavelength band?
Advertisements
Solution
A radio can tune to minimum frequency, v1 = 7.5 MHz = 7.5 × 106 Hz
Maximum frequency, v2 = 12 MHz = 12 × 106 Hz
Speed of light, c = 3 × 108 m/s
Corresponding wavelength for v1 can be calculated as:
`lambda_1 = "c"/"v"_1`
= `(3 xx 10^8)/(7.5 xx 10^6)`
= 40 m
Corresponding wavelength for v2 can be calculated as:
`lambda_2 = "c"/"v"_2`
= `(3 xx 10^8)/(12 xx 10^6)`
= 25 m
Thus, the wavelength band of the radio is 40 m to 25 m.
APPEARS IN
RELATED QUESTIONS
Give a reason for the following:
It is necessary to use satellites for long-distance TV transmission. Why?
Which radiation is used for satellite communication?
Name the region beyond the red end of the spectrum.
Name the radiations of wavelength just longer than 8 × 10-7m.
Name the radiations of wavelength just shorter than 4 × 10-7 m.
Two waves A and B have wavelength 0.01 Å and 9000 Å respectively.
- Name the two waves.
- Compare the speeds of these waves when they travel in vacuum.
Name three properties of ultraviolet radiations which are similar to visible light.
In a Coolidge tube, electrons strike the target and stop inside it. Does the target get more and more negatively charged as time passes?
The Kα and Kβ X-rays of molybdenum have wavelengths 0.71 A and 0.63 A respectively. Find the wavelength of Lα X-ray of molybdenum.
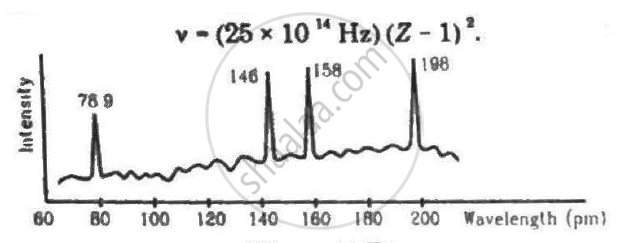
Continuous X-rays are made to strike a tissue paper soaked with polluted water. The incoming X-rays excite the atoms of the sample by knocking out the electrons from the inner shells. Characteristic X-rays are analysed and the intensity is plotted against the wavelength. Assuming that only Kα intensities are detected, list the elements present in the sample from the plot. Use Moseley's equation v − (25 × 1014Hz)(Z − 1)2.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Write the range of the wavelength of the following electromagnetic radiations:
(a) Infrared rays
(b) Ultraviolet rays
(c) γ -rays
Write one use of each of the above.
To which regions of the electromagnetic spectrum do the following wavelengths belong:
(a) 250 nm
(b) 1500 nm
Answer briefly.
Why are microwaves used in radar?
Answer briefly.
Give two uses of ultraviolet rays.
Answer briefly.
Why high-frequency carrier waves are used for the transmission of audio signals?
Solve the numerical problem.
Calculate the wavelength in nm of an X-ray wave of frequency 2.0 × 1018 Hz.
For television broadcasting, the frequency employed is normally
Find the photon energy in units of ev for electromagnetic wave if waves length 40 m. Given h = 6.63 × 10–34 J.
The electric field intensity produced by the radiations coming from 100 W bulb at a 3 m distance is E. The electric field intensity produced by the radiations coming from 50 W bulb at the same distance is ______.
Electromagnetic waves with wavelength
- λ1 is used in satellite communication.
- λ2 is used to kill germs in water purifies.
- λ3 is used to detect leakage of oil in underground pipelines.
- λ4 is used to improve visibility in runways during fog and mist conditions.
- Identify and name the part of electromagnetic spectrum to which these radiations belong.
- Arrange these wavelengths in ascending order of their magnitude.
- Write one more application of each.
