Science (English Medium)
Academic Year: 2024-2025
Date & Time: 27th February 2025, 10:30 am
Duration: 3h
Advertisements
General Instructions:
Read the following instructions carefully and follow them:
- This question paper contains 33 questions. All questions are compulsory.
- This question paper is divided into five sections - Section A, B, C, D and E.
- Section A - questions number 1 to 16 are multiple choice type questions. Each question carries 1 mark.
- Section B - questions number 17 to 21 are very short answer type questions. Each question carries 2 marks.
- Section C - questions number 22 to 28 are short answer type questions. Each question carries 3 marks.
- Section D - questions number 29 and 30 are case-based questions. Each question carries 4 marks.
- Section E - questions number 31 to 33 are long answer type questions. Each question carries 5 marks.
- There is no overall choice given in the question paper. However, an internal choice has been provided in few questions in all the sections except Section - A.
- Kindly note that there is a separate question paper for Visually Impaired candidates.
- Use of calculator is NOT allowed.
You may use the following values of physical constants wherever necessary:
c = 3 × 108 m/s
h = 6.63 × 10−34 Js
e = 1.6 × 10−19 C
µ0 = 4π × 10−7 T m A−1
ε0 = 8.854 × 10−12 C2 N−1 m−2
`1/(4πε_0)` = 9 × 109 N m2 C−2
Mass of electron (me) = 9.1 × 10−31 kg
Mass of neutron = 1.673 × 10−27 kg
Avogadro’s number = 6.023 × 1023 per gram mole
Boltzmann’s constant = 1.38 × 10−23 JK−1
Assertion: All naturally occurring α-aminoacids except glycine are optically active.
Reason: Most naturally occurring amino acids have L-configuration.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false and Reason (R) is true.
Chapter:
Assertion (A): The boiling points of ethanolis higher than that of methoxymethane.
Reason (R): There is intramolecular hydrogen bonding in ethanol.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false and Reason (R) is true.
Chapter:
Assertion: The boiling points of alkyl halides decrease in the order:
RI > RBr > RCl > RF
Reason: The boiling points of alkyl chlorides, bromides and iodides are considerably higher than that of the hydrocarbon of comparable molecular mass.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false and Reason (R) is true.
Chapter:
Assertion (A): [Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2 are example of homoleptic complexes.
Reason (R): All the ligands attached to the metal are the same.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false and Reason (R) is true.
Chapter:
An unripe mango placed in a concentrated salt solution to prepare pickle, shrivels because ______.
it gains water due to osmosis
it loses water due to reverse osmosis
it gains water due to reverse osmosis
it loses water due to osmosis
Chapter: [1] Solutions
Which of the following statements is not true about glucose?
It is an aldohexose.
On heating with HI, it forms n-hexane.
It exists in furanose form.
It does not give Schiff’s test.
Chapter:
The best reagent for converting propanamide into propanamine is ______.
excess H2
Br2 in aqueous NaOH
iodine in the presence of red phosphorus
LiAlH4 in ether
Chapter:
The acid formed when propyl magnesium bromide is treated with CO2 followed by acid hydrolysis is ______.
C3H7COOH
C2H5COOH
CH3COOH
C3H7OH
Chapter:
Which is the correct order of acid strength from the following?
C6H5OH > H2O > ROH
C6H5 > ROH > H2O
ROH > C6H5OH > ROH
H2O > C6H5OH > ROH
Chapter:
Alkyl halides undergoing nucleophilic bimolecular substitution reaction involve ______.
retention of configuration
formation of racemic mixture
inversion of configuration
formation of carbocation
Chapter:
Arrange the following compounds in increasing order of their boiling points.
(a) \[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
\backslash\phantom{...........}\\
\ce{CH - CH - CH2Br}\\
/\phantom{.............}\\
\ce{CH3}\phantom{..................}
\end{array}\]
(b) \[\ce{CH3CH2CH2CH2Br}\]
(c) \[\begin{array}{cc}
\phantom{...}\ce{CH3}\\
\phantom{}|\\
\ce{H3C - C - CH3}\\
\phantom{}|\\
\phantom{..}\ce{Br}
\end{array}\]
(b) < (a) < (c)
(a) < (b) < (c)
(c) < (a) < (b)
(c) < (b) < (a)
Chapter: [6] Haloalkanes and Haloarenes
The correct IUPAC name of [Pt(NH3)2Cl2]2+ is______.
Diamminedichloridoplatinum (II)
Diamminedichloridoplatinum (IV)
Diamminedichloridoplatinum (O)
Diamminedichloridoplatinate (IV)
Chapter:
Acidified KMnO4 oxidises sulphite to ______.
\[\ce{S_2O^2-_4}\]
\[\ce{S_2O^2-_8}\]
SO2(g)
\[\ce{SO^2-_4}\]
Chapter:
The magnetic moment is associated with its spin angular momentum and orbital angular momentum. Spin only magnetic moment value of \[\ce{Cr^{3+}}\] ion is ______.
2.87 B.M.
3.87 B.M.
3.47 B.M.
3.57 B.M.
Chapter: [4] d-block and f-block Elements
Standard electrode potential for Sn+4/Sn+2 couple is +0.15 V, and that for the Cr+3/Cr couple is –0.74 V. The two couples in their standard states are connected to make a cell. The cell potential will be ______.
+1.19 V
+0.89 V
+0.18 V
+1.83 V
Chapter:
In case of association, abnormal molar mass of solute will ______.
increase
decrease
remain same
first increase and then decrease
Chapter:
Identify A and B in each of the following reaction sequencе:
\[\ce{CH3CH2Cl ->[NaCN][]A->[H2/Ni][]B}\]
Chapter:
Identify A and B in each of the following reaction sequencе:
\[\ce{C6H5NH2 ->[NaNO2/HCl][0.5°C]A->[C6H5NH2][H+]B}\]
Chapter:
When FeCr2O4 is fused with Na2CO3 in the presence of air it gives a yellow solution of compound (А). Compound (A) on acidification gives compound (B). Compound (B) on reaction with KCl forms an orange coloured (C). An acidified solution of compound (C) oxidises iodide to (D). Identify (A), (B), (C) and (D).
Chapter:
Would you expect benzaldehyde to be more reactive or less reactive in nucleophilic addition reactions than propanal? Justify your answer.
Chapter:
Advertisements
Give reason:
Cooking is faster in a pressure cooker than in an open pan.
Chapter:
Given reason:
On mixing liquid X and liquid Y, the volume of the resulting solution decreases. What type of deviation from Raoult’s law is shown by the resulting solution? What change in temperature would you observe after mixing liquids X and Y?
Chapter:
What type of azeotrope is formed by positive deviation from Raoult’s law?
Chapter: [1] Solutions
Give reasons for the following:
The melting points of α-amino acids are generally higher than that of the corresponding carboxylic acids.
Chapter:
Give reasons for the following:
Amino acids show amphoteric behaviour.
Chapter:
A solution containing 15 g urea (molar mass = 60 g mol–1) per litre of solution in water has the same osmotic pressure (isotonic) as a solution of glucose (molar mass = 180 g mol–1) in water. Calculate the mass of glucose present in one litre of its solution.
Chapter: [1] Solutions
Calculate ΔrG° and log KC of the reaction:
\[\ce{Fe{^{2+}_{(aq)}} + Ag{^{+}_{(aq)}} -> Fe{^{3+}_{(aq)}} + Ag_{(s)}}\]
Given `E_(Ag^+//Ag)^° = 0.80 V, E_(Fe^(3+)//Fe^(2+))^° = 0.77 V`
[R = 8.314 J K−1, F = 96500 C mol−1]
Chapter:
Arrange the following in decreasing order of pKb:
Aniline, p-nitroaniline, p-methylaniline
Chapter:
Account for the following.
Diazonium salts of aromatic amines are more stable than those of aliphatic amines.
Chapter:
Account for the following:
Methylamine in water reacts with ferric chloride to precipitate hydrated ferric oxide.
Chapter: [9] Amines
Draw the structure of the major monohalo product of the following reaction:
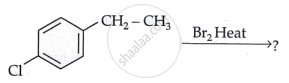
Chapter:
Draw the structure of the major monohalo product for the following reaction:

Chapter:
Draw the structure of the major monohalo product of the following reaction:
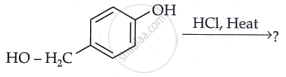
Chapter:
How are the following conversions carried out?
propene to 1-iodopropane
Chapter:
How do you convert:
2-bromobutane to but-2-ene
Chapter: [6] Haloalkanes and Haloarenes
The elements of 3d transition series are given as:
Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn
Answer the following:
Copper has exceptionally positive `E_(M^(2+)//M)^°` value, why?
Chapter:
The elements of 3d transition series are given as:
Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn
Answer the following:
Which element is a strong reducing agent in +2 oxidation state and why?
Chapter:
Advertisements
The elements of 3d transition series are given as:
Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn
Answer the following:
Zn2+ salts are colourless. Why?
Chapter:
A certain reaction is 50% complete in 20 minutes at 300 K and the same reaction is 50% complete in 5 minutes at 350 K. Calculate the activation energy if it is a first order reaction.
[R = 8.314 J K−1 mol−1, log 4 = 0.602]
Chapter:
Write the product obtained when D-glucose reacts with H2N − OH.
Chapter: [10] Biomolecules
Write the reaction involved when D-glucose is treated with the following reagent:
(CH3CO)2O
Chapter: [10] Biomolecules
Why vitamin C cannot be stored in our body?
Chapter: [10] Biomolecules
The following questions are case based questions. Read the passage carefully and answer the questions that follow.
| Phenols undergo electrophilic substitution reactions readily due to the strong activating effect of OH group attached to the benzene ring. Since, the OН group increases the electron density more to oand p-positions, OH group is ortho, para-directing. Reimer-Tiemann reaction is one of the examples of aldehyde group being introduced on the aromatic ring of phenol, ortho to the hydroxyl group. This is a general method used for the ortho-formylation of phenols. |
Answer the following questions:
- What happens when phenol reacts with [2]
- Br2/CS2
- Conc. HNO3
- Why does phenol not undergo protonation readily? [1]
- Which is a stronger acid-phenol or cresol? Give reasons. [1]
OR
Write the IUPAC name of the product formed in the Reimer-Tiemann reaction. [1]
Chapter:
The following questions are case based questions. Read the passage carefully and answer the questions that follow.
| The rate of a chemical reaction is expressed either in terms of decrease in the concentration of reactants or increase in the concentration of a product per unit time. The rate of the reaction depends upon the nature of reactants, concentration of reactants, temperature, presence of catalyst, surface area of the reactants and presence of light. The rate of reactions is directly related to the concentration of reactant. Rate law states that the rate of reaction depends upon the concentration terms on which the rate of reaction actually depends, as observed experimentally. The sum of powers of the concentration of the reactants in the Rate law expression is called the order of reaction, while the number of reacting species taking part in an elementary reaction which must collide simultaneously, in order to bring about a chemical reaction is called molecularity of the reaction. |
- Answer the following questions: [2]
- What is a rate determining step?
- Define complex reaction.
- What is the effect of temperature on the rate constant of a reaction? [1]
OR
Why is molecularity applicable only for elementary reactions whereas order is applicable for elementary as well as complex reactions? [1] - The conversion of molecule X to Y follows second order kinetics. If concentration of X is increased threе times, how will it affect the rate of formation of Y? [1]
Chapter:
How will you convert ethanal into the following compound?
But-2-enal
Chapter: [8] Aldehydes, Ketones and Carboxylic Acids
Carry out the following conversion:
Propanoic acid to ethane
Chapter:
An alkene A with molecular formula C5H10 on ozonolysis gives a mixture of two compounds B and C. Compound B gives positive Fehling test and also reacts with iodine and NaOH solution. Compound C does not give Fehling solution test but forms iodoform. Identity the compounds A, B and C.
Chapter:
An organic compound (A) (molecular formula C8H16O2) was hydrolysed with dilute sulphuric acid to give a carboxylic acid (B) and an alcohol (C). Oxidation of (C) with chromic acid produced (B). (C) on dehydration gives but-1-ene. Write equations for the reactions involved.
Chapter: [8] Aldehydes, Ketones and Carboxylic Acids
For the complex [Fe(en)2Cl2]Cl, identify: [5]
- the oxidation number of iron.
- the hybridisation and the shape of the complex.
- the magnetic behaviour of the complex.
- whether there is an optical isomer of the complex? If so draw its structure.
- IUPAC name of the complex. (At. no. of Fe = 26)
Chapter:
Using the IUPAC norm, write the systematic name of the following:
[Co(NH3)4Cl(NO2)]Cl
Chapter:
Write the IUPAC name of the following coordination compound:
K3[Fe(CN)6]
Chapter:
Using IUPAC norm, write the name of the following:
[Cr(C2O4)3]3−
Chapter:
What is crystal field splitting energy?
Chapter: [5] Coordination Compounds
Why are low spin tetrahedral complexes not formed?
Chapter: [5] Coordination Compounds
Write the cell reaction and calculate the e.m.f of the following cell at 298 K:
Sn(s) | Sn2+ (0.004 M) || H+ (0.020 M) | H2(g) (1 bar) | Pt(s)
(Given: `E_(Sn^(2+)"/"Sn)^° = -0.14` V)
Chapter: [2] Electrochemistry
Give reasons:
On the basis of E° values, O2 gas should be liberated at anode but it is Cl2 gas which is liberated in the electrolysis of aqueous NaCl.
Chapter: [2] Electrochemistry
Give reasons:
Conductivity of CH3COOH decreases on dilution.
Chapter: [2] Electrochemistry
Write the cell reactions which occur in lead storage battery when the battery is in use.
Chapter:
Calculate the potential for half-cell containing 0.01 M K2Cr2O7(aq), 0.01 M Cr3+(aq) and 1.0 × 10−4 M H+(aq). The half cell reaction is
\[\ce{Cr_2O^{2-}_7 + 14 H+_{(aq)} + 6e -> 2Cr^{3+}_{(aq)} + 7H2O_{(l)}}\]
and the standard electrode potential is given as E° = 1.33 V.
[Given: log 10 = 1]
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CBSE previous year question papers Class 12 Chemistry with solutions 2024 - 2025
Previous year Question paper for CBSE Class 12 -2025 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CBSE Class 12.
How CBSE Class 12 Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
