Advertisements
Advertisements
प्रश्न
Assertion (A): The boiling points of ethanolis higher than that of methoxymethane.
Reason (R): There is intramolecular hydrogen bonding in ethanol.
पर्याय
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false and Reason (R) is true.
MCQ
विधान आणि तर्क
Advertisements
उत्तर
Assertion (A) is true, but Reason (R) is false.
Explanation:
The boiling point of ethanol exceeds that of methoxymethane owing to intermolecular hydrogen bonding rather than intramolecular hydrogen bonding.
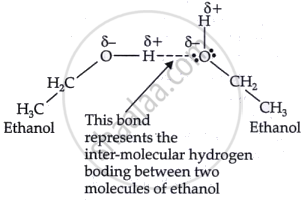
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
