Advertisements
Advertisements
प्रश्न
The following questions are case based questions. Read the passage carefully and answer the questions that follow.
| Phenols undergo electrophilic substitution reactions readily due to the strong activating effect of OH group attached to the benzene ring. Since, the OН group increases the electron density more to oand p-positions, OH group is ortho, para-directing. Reimer-Tiemann reaction is one of the examples of aldehyde group being introduced on the aromatic ring of phenol, ortho to the hydroxyl group. This is a general method used for the ortho-formylation of phenols. |
Answer the following questions:
- What happens when phenol reacts with [2]
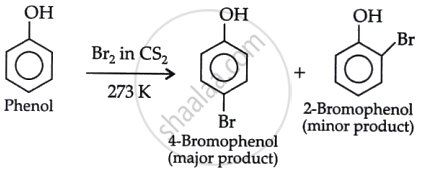
- Br2/CS2
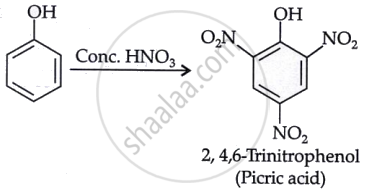
- Conc. HNO3
- Why does phenol not undergo protonation readily? [1]
- Which is a stronger acid-phenol or cresol? Give reasons. [1]
OR
Write the IUPAC name of the product formed in the Reimer-Tiemann reaction. [1]
घटनेचा अभ्यास
Advertisements
उत्तर
(a)
- Phenol + Br2/CS2:
- Phenol + conc. HNO3:
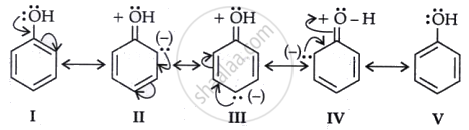
(b) One pair of oxygen atoms in phenol resonates with the benzene ring, giving oxygen a positive charge. It will not be easy for it to become protonated.
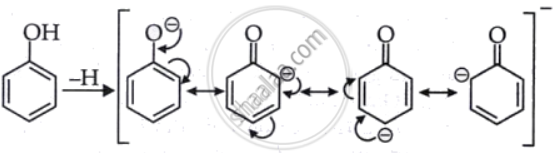
(c) An acid that is stronger is phenol. Because of the +I effect and the hyper-conjugation effect of the methyl group in cresol, there are more electrons pointing toward the oxygen atom, which makes the conjugate base less stable. The phenoxide ion is resonance stabilized in phenol, which makes it a stronger acid than cresol.
OR
The product is salicylaldehyde IUPAC name is 2-Hydroxybenzaldehyde.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
2024-2025 (March) Delhi Set 1
