Advertisements
Advertisements
A photographic film is coated with a silver bromide layer. When light falls on this film, silver bromide molecules dissociate and the film records the light there. A minimum of 0.6 eV is needed to dissociate a silver bromide molecule. Find the maximum wavelength of light that can be recorded by the film.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Concept: undefined >> undefined
In an experiment on photoelectric effect, light of wavelength 400 nm is incident on a cesium plate at the rate of 5.0 W. The potential of the collector plate is made sufficiently positive with respect to the emitter, so that the current reaches its saturation value. Assuming that on average, one out of every 106 photons is able to eject a photoelectron, find the photocurrent in the circuit.
Concept: undefined >> undefined
Advertisements
The first excited energy of a He+ ion is the same as the ground state energy of hydrogen. Is it always true that one of the energies of any hydrogen-like ion will be the same as the ground state energy of a hydrogen atom?
Concept: undefined >> undefined
Which wavelengths will be emitted by a sample of atomic hydrogen gas (in ground state) if electrons of energy 12.2 eV collide with the atoms of the gas?
Concept: undefined >> undefined
A light beam of wavelength 400 nm is incident on a metal plate of work function 2.2 eV. (a) A particular electron absorbs a photon and makes two collisions before coming out of the metal. Assuming that 10% of the extra energy is lost to the metal in each collision, find the kinetic energy of this electron as it comes out of the metal. (b) Under the same assumptions, find the maximum number of collisions the electron can suffer before it becomes unable to come out of the metal.
Concept: undefined >> undefined
When white radiation is passed through a sample of hydrogen gas at room temperature, absorption lines are observed in Lyman series only. Explain.
Concept: undefined >> undefined
What will be the energy corresponding to the first excited state of a hydrogen atom if the potential energy of the atom is taken to be 10 eV when the electron is widely separated from the proton? Can we still write En = E1/n2, or rn = a0 n2?
Concept: undefined >> undefined
A horizontal cesium plate (φ = 1.9 eV) is moved vertically downward at a constant speed v in a room full of radiation of wavelength 250 nm and above. What should be the minimum value of v so that the vertically-upward component of velocity is non-positive for each photoelectron?
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Concept: undefined >> undefined
The minimum orbital angular momentum of the electron in a hydrogen atom is
Concept: undefined >> undefined
Figure shows a straight, long wire carrying a current i and a rod of length l coplanar with the wire and perpendicular to it. The rod moves with a constant velocity v in a direction parallel to the wire. The distance of the wire from the centre of the rod is x. Find the motional emf induced in the rod.
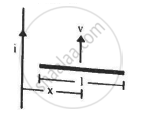
Concept: undefined >> undefined
In which of the following transitions will the wavelength be minimum?
Concept: undefined >> undefined
In which of the following systems will the radius of the first orbit (n = 1) be minimum?
Concept: undefined >> undefined
Which of the following curves may represent the speed of the electron in a hydrogen atom as a function of trincipal quantum number n?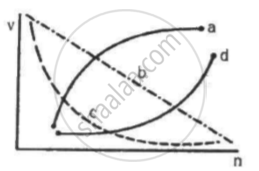
Concept: undefined >> undefined
In the arrangement shown in the figure, y = 1.0 mm, d = 0.24 mm and D = 1.2 m. The work function of the material of the emitter is 2.2 eV. Find the stopping potential V needed to stop the photocurrent.
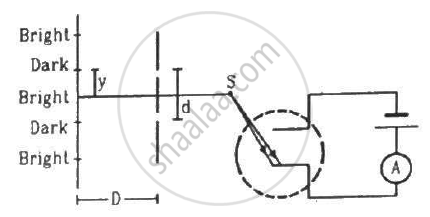
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Concept: undefined >> undefined
As one considers orbits with higher values of n in a hydrogen atom, the electric potential energy of the atom
Concept: undefined >> undefined
In an experiment on photoelectric effect, the emitter and the collector plates are placed at a separation of 10 cm and are connected through an ammeter without any cell. A magnetic field B exists parallel to the plates. The work function of the emitter is 2.39 eV and the light incident on it has wavelengths between 400 nm and 600 nm. Find the minimum value of B for which the current registered by the ammeter is zero. Neglect any effect of space charge.
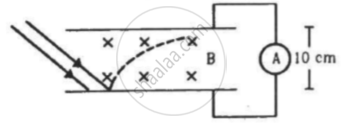
Concept: undefined >> undefined
The radius of the shortest orbit in a one-electron system is 18 pm. It may be
Concept: undefined >> undefined
A silver ball of radius 4.8 cm is suspended by a thread in a vacuum chamber. Ultraviolet light of wavelength 200 nm is incident on the ball for some time during which light energy of 1.0 × 10−7 J falls on the surface. Assuming that on average, one photon out of every ten thousand is able to eject a photoelectron, find the electric potential at the surface of the ball, assuming zero potential at infinity. What is the potential at the centre of the ball?
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Concept: undefined >> undefined
A hydrogen atom in ground state absorbs 10.2 eV of energy. The orbital angular momentum of the electron is increased by
Concept: undefined >> undefined
An electron with kinetic energy 5 eV is incident on a hydrogen atom in its ground state. The collision
Concept: undefined >> undefined
