Advertisements
Advertisements
Question
Which of the following curves may represent the speed of the electron in a hydrogen atom as a function of trincipal quantum number n?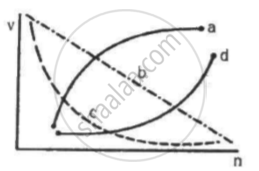
Advertisements
Solution
(c)
The speed (v) of electron can be expressed as
`v = (Ze^2)/(2∈_0hn)` ....(1)
Here,
Z = Number of protons in the nucleus
e = Magnitude of charge on electron charge
n = Principal quantum number
h = Planck's constant
It can be observed from equation (1) that the velocity of electron is inversely proportional to the principal quantum number (n).
Therefore, the graph between them must be a rectangular hyperbola.
The correct curve is (c).
APPEARS IN
RELATED QUESTIONS
Which wavelengths will be emitted by a sample of atomic hydrogen gas (in ground state) if electrons of energy 12.2 eV collide with the atoms of the gas?
When white radiation is passed through a sample of hydrogen gas at room temperature, absorption lines are observed in Lyman series only. Explain.
The minimum orbital angular momentum of the electron in a hydrogen atom is
In which of the following transitions will the wavelength be minimum?
Ionization energy of a hydrogen-like ion A is greater than that of another hydrogen-like ion B. Let r, u, E and L represent the radius of the orbit, speed of the electron, energy of the atom and orbital angular momentum of the electron respectively. In ground state
Find the binding energy of a hydrogen atom in the state n = 2.
(a) Find the first excitation potential of He+ ion. (b) Find the ionization potential of Li++ion.
A hydrogen atom in a state having a binding energy of 0.85 eV makes transition to a state with excitation energy 10.2 e.V (a) Identify the quantum numbers n of the upper and the lower energy states involved in the transition. (b) Find the wavelength of the emitted radiation.
Whenever a photon is emitted by hydrogen in Balmer series, it is followed by another photon in Lyman series. What wavelength does this latter photon correspond to?
A hydrogen atom in state n = 6 makes two successive transitions and reaches the ground state. In the first transition a photon of 1.13 eV is emitted. (a) Find the energy of the photon emitted in the second transition (b) What is the value of n in the intermediate state?
Find the maximum angular speed of the electron of a hydrogen atom in a stationary orbit.
The average kinetic energy of molecules in a gas at temperature T is 1.5 kT. Find the temperature at which the average kinetic energy of the molecules of hydrogen equals the binding energy of its atoms. Will hydrogen remain in molecular from at this temperature? Take k = 8.62 × 10−5 eV K−1.
Electrons are emitted from an electron gun at almost zero velocity and are accelerated by an electric field E through a distance of 1.0 m. The electrons are now scattered by an atomic hydrogen sample in ground state. What should be the minimum value of E so that red light of wavelength 656.3 nm may be emitted by the hydrogen?
A hydrogen atom moving at speed υ collides with another hydrogen atom kept at rest. Find the minimum value of υ for which one of the atoms may get ionized.
The mass of a hydrogen atom = 1.67 × 10−27 kg.
The Balmer series for the H-atom can be observed ______.
- if we measure the frequencies of light emitted when an excited atom falls to the ground state.
- if we measure the frequencies of light emitted due to transitions between excited states and the first excited state.
- in any transition in a H-atom.
- as a sequence of frequencies with the higher frequencies getting closely packed.
Positronium is just like a H-atom with the proton replaced by the positively charged anti-particle of the electron (called the positron which is as massive as the electron). What would be the ground state energy of positronium?
