Definitions [1]
Define the following term:
Anomers
Anomers are stereoisomers of sugars that differ only in the configuration of the hydroxyl group at the C1 position. They are not mirror images of each other.
Theorems and Laws [2]
Alfred Werner (1893) proposed the first systematic theory to explain the structure and bonding in coordination compounds. His key postulates are:
Main Postulates:
- In coordination compounds, metals show two types of valencies — Primary (ionisable) and Secondary (non-ionisable).
- Primary valences are normally ionisable. They are satisfied by negative ions (counter ions/anions). They correspond to the metal's oxidation state.
- Secondary valences are non-ionisable. They are satisfied by neutral molecules or negative ions (ligands). The secondary valency is equal to the coordination number and is constant for a metal.
- Ion groups bound by secondary valencies to the metal have a characteristic spatial arrangement (geometry). This geometry is decided by the secondary valences, not the primary valences.
Werner's Formula Examples:
- CrCl₃·6H₂O: In [Cr(H₂O)₆]Cl₃, all three Cl⁻ are outside the coordination sphere and hence ionisable.
- CrCl₃·5H₂O: [Cr(H₂O)₅Cl]Cl₂ — two Cl⁻ are ionisable, one is inside the coordination sphere.
- CrCl₃·4H₂O: [Cr(H₂O)₄Cl₂]Cl — one Cl⁻ ionisable.
- CrCl₃·3H₂O: [Cr(H₂O)₃Cl₃] — no ionisable Cl⁻, no precipitate with AgNO₃.
- Proposed by Heitler and London (1927), further developed by Pauling and Slater.
- A covalent bond is formed when half-filled valence atomic orbitals of similar energies overlap, each containing one unpaired electron.
- Greater the overlap → stronger the bond.
Types of Orbital Overlap:
| Type | Description | Bond Formed |
|---|---|---|
| Axial (Head-on) overlap | Orbitals overlap along the internuclear axis | Sigma (σ) bond |
| Sidewise (Lateral) overlap | Orbitals overlap parallel to each other, perpendicular to the internuclear axis | Pi (π) bond |
Hybridisation & Shapes:
| Hybridisation | Shape | Coordination No. |
|---|---|---|
| sp³ | Tetrahedral | 4 |
| dsp² | Square planar | 4 |
| sp³d | Trigonal bipyramidal | 5 |
| d²sp³ | Octahedral (inner) | 6 |
| sp³d² | Octahedral (outer) | 6 |
Limitations of VBT:
- Involves a number of assumptions.
- Does not give a quantitative interpretation of magnetic data.
- Does not explain the colour exhibited by coordination compounds.
- Does not give a quantitative interpretation of the thermodynamic or kinetic stabilities of coordination compounds.
- Does not make exact predictions regarding the tetrahedral and square planar structures of 4-coordinate complexes.
- Does not distinguish between weak and strong ligands.
Key Points
Ligands:
Ligands are the donor atoms, molecules, or anions that donate a pair of electrons to the metal atom or ion and form a coordinate bond. The number of coordinating or ligating groups present in a ligand is called the denticity of that ligand.
| Type | Description | Examples |
|---|---|---|
| (i) Unidentate / Monodentate | Binds through one donor atom | Cl⁻, H₂O, NH₃, NO |
| (ii) Didentate / Bidentate | Binds through two donor atoms | en (ethane-1,2-diamine), C₂O₄²⁻ (oxalate) |
| (iii) Polydentate | Several donor atoms in a single ligand | N(CH₂CH₂NH₂)₃ |
| (iv) Ambidentate | Two different donor atoms, either of which can coordinate | NO₂⁻ (through N or O), SCN⁻ (through S or N), NCS⁻ |
| (v) Chelating | Forms a ring structure with the central atom; polydentate chelate complexes are more stable than monodentate analogues | EDTA (hexadentate), en (bidentate), C₂O₄²⁻ |
NH₄⁺ is NOT a ligand — the N atom in NH₄⁺ has no lone pair of electrons to donate (lone pair has been donated to H⁺). Thiosulphato (S₂O₃²⁻) is an ambidentate ligand, NOT a chelating ligand.
Coordination Number:
The coordination number (CN) of a metal ion in a complex is the total number of unidentate ligands (plus double the number of didentate ligands if any) attached to the central metal ion through coordinate bonds.
Homoleptic vs Heteroleptic Complexes:
- Homoleptic complexes: Metal is bound to only one kind of donor group. e.g., [Co(NH₃)₆]³⁺
- Heteroleptic complexes: Metal is bound to more than one kind of donor group. e.g., [Co(NH₃)₄Cl₂]⁺
- In coordination compounds, d-orbitals split into t₂g (lower) and eg (higher) energy levels due to the ligand field.
- The energy difference between them is called the crystal field splitting energy (Δ₀).
- This Δ₀ lies in the visible region, so these compounds absorb visible light.
- When light is absorbed, an electron jumps from t₂g → eg, called a d–d transition.
- The observed colour is complementary to the colour of light absorbed.
- The energy relation is: E = hν = Δ₀.
- Metal ions with d¹–d⁹ configuration are coloured, while d⁰ and d¹⁰ are colourless.
- Some compounds (e.g., KMnO₄) show colour due to charge transfer (LMCT), not d–d transition.
- Ligand strength affects colour: strong field ligands ↑ Δ₀, weak ligands ↓ Δ₀.
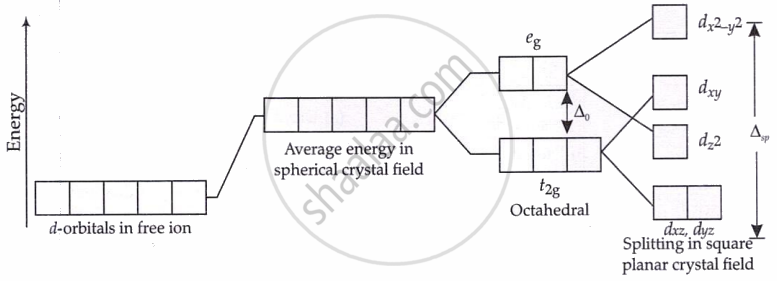
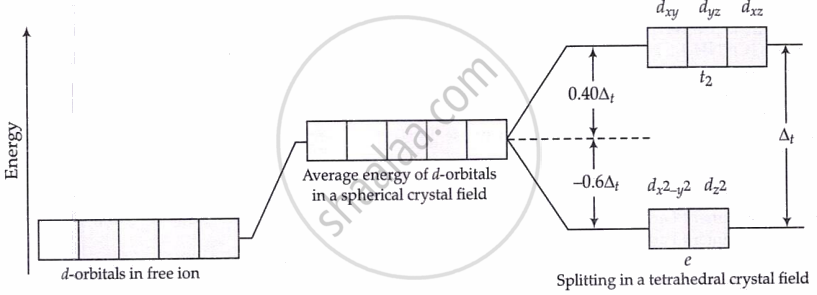
- Geometry affects splitting: tetrahedral complexes have smaller splitting
Δₜ = 4/9 Δ₀ (e.g., Co²⁺: pink → blue change).
CFT is an electrostatic model that considers the metal-ligand bond to be ionic, arising purely from electrostatic interactions between the metal ion and the ligand (treated as point charges for anions, or point dipoles for neutral molecules).
CFT considers the effect of ligands on the relative energies of the d-orbitals of the central metal atom/ion.
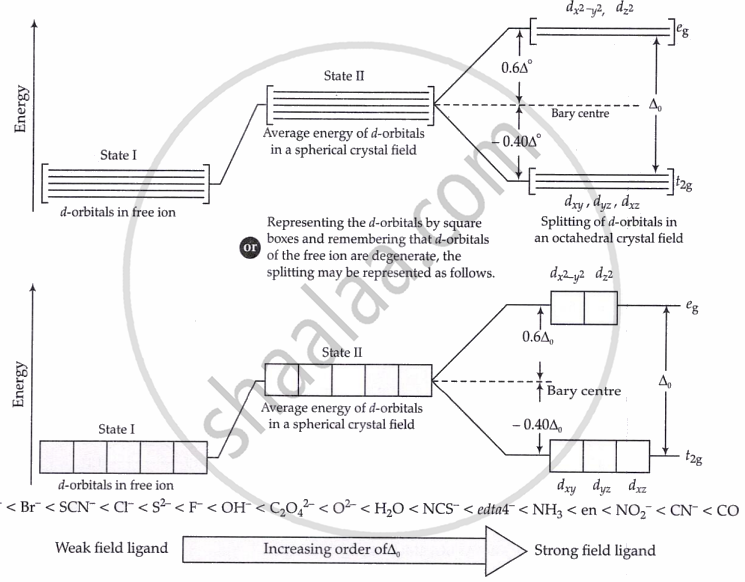
If Δ₀ < P, 4th electron will enter eg giving the configuration \[t_{2g}^3e_{g}^1.\] Ligands for which Δ₀ < P are called weak field ligands.
If Δ₀ > P, pairing will occur in the t2g orbitals and eg orbitals will remain vacant. So, the configuration for 4th e⁻ will be \[t_{2g}^4e_{g}^0.\]. For Δ₀ > P, ligands are strong field ligands.
Splitting of d-orbitals in a square planar crystal field:
Splitting of d-orbital in a tetrahedral crystal field:
Important Questions [34]
- Write ‘four’ characteristics of co-ordinate complex ion.
- Illustrate with Example, the Difference Between a Double Salt and a Co-ordination Compound
- What is Effective Atomic Number? Calculate Effective Atomic Number of Copper
- How Ligands Are Classified? Explain with Suitable Examples.
- Explain Sidgwick’s electronic theory with suitable example
- Explain the following, giving two examples: Heteroleptic
- Classify the Following Ligands into Monodentate and Polydentate
- Write the structure and IUPAC names of all the metamers represented by formula C4H10
- Which of the Following Compounds is Used as a Semipermeable Membrane?
- Predict the Co-ordination No. of Cs+ Ion
- Iupac Name of the Following Compound is
- Write Iupac Names of the Following Compounds 3-methylcyclohexanone
- Write Iupac Names of the Following Compounds: Butane-2,3-dione
- How is Benzonitrile Converted to Benzophenone?
- Write Applications of Co-ordination Compounds in Medicine and Electroplating.
- The Ligand Triethylenetetramine is _______.
- Explain the following, giving two examples: Homoleptic
- Which of the Following Complexes Will Give a White Precipitate on Treatment with a Solution of Barium Nitrate?
- Explain Optical Activity
- Explain Optical Activity of Lactic Acid
- Write Four Postulates of Werner'S Theory.
- Mohr’s Salt is Ferrous ammonium sulphate
- On the basis of valence bond theory explain the nature of bonding in [CoF6]3 ion.
- Explain the Geometry of `[Co(Nh_3)_6]^(3+)` On the Basis of Hybridisation. (Z of Co = 27)
- State the superiority of crystal field theory over valence bond theory.
- How Are the Following Conversions Carried Out? Benzoic Acid into Metanitrobenzoic Acid.
- Explain cationic complexes and anionic complexes of co-ordination compounds.
- The complex ion[Co(H2O)5 (ONO)]^(2+)
- What is ‘Effective Atomic Number’ (Ean)?
- Calculate the Effective Atomic Number of the Central Metal Atom in the Following Compounds
- Effective Atomic Number Rule is Used to Find
- Calculate EAN of cobalt (Z=27) in [CO(NH3)6]+3 and of zinc (Z=30) in [Zn(NH3)4]SO4.
- What is the Action of Lithium Aluminum Hydride in the Presence of Ether on the Following Compounds? A. Nitroethane B. 2-methyl-1-nitropropane
- Calculate the Effective Atomic Number of the Central Metal Atom in the Following Compounds: Cr(Co)6 Cr(Z=24)
Concepts [12]
- Introduction
- Importance of Coordination Compounds
- Important Terms Pertaining to Coordination Compounds
- Colour in Coordination Compounds
- Magnetic Properties of Coordination Compounds
- Bonding in Coordination Compounds
- Werner’s Theory of Coordination Compounds
- Valence Bond Theory (VBT)
- Crystal Field Theory (CFT)
- Stereoisomerism
- Structural Isomerism
- Importance of Coordination Compounds
