Advertisements
Advertisements
Question
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - O - CH2 - CH - CH3}\\
\phantom{..........}|\\
\phantom{............}\ce{CH3}
\end{array}\]
Advertisements
Solution
1-Methoxy-2-methylpropane
APPEARS IN
RELATED QUESTIONS
Name the following compound according to IUPAC system.

Give IUPAC name of the following ether:
\[\begin{array}{cc}
\ce{C2H5OCH2 - CH - CH3}\\
\phantom{.....}|\\
\phantom{.......}\ce{CH3}
\end{array}\]
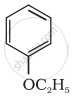
Give IUPAC name of the following ether:
Write the structures of the products when Butan-2-ol reacts with CrO3
Write structural formula for pentane-1,4-diol.
Glycerol is ____________.
Give IUPAC names of the following compound:
\[\begin{array}{cc}
\phantom{..}\ce{H}\phantom{...}\ce{CH3}\phantom{.}\ce{H}\phantom{..}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - H}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\phantom{.}\ce{H}\phantom{...}\ce{OH}\phantom{.}\ce{H}\phantom{.}\\
\end{array}\]
Butane-2-ol is ____________.
Which of the following compounds is oxidised to prepare methyl ethyl ketone?
Which of the following is most acidic?
The heating of phenyl methyl ether with HI produces:
Which of the following gives a positive iodoform test?
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
IUPAC name of m-cresol is ______.
Explain why p-nitrophenol is more acidic than phenol.
Assertion: Addition reaction of water to but-1-ene in acidic medium yields butan-1-ol.
Reason: Addition of water in acidic medium proceeds through the formation of primary carbocation.
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
How can phenol be converted to aspirin?
