Advertisements
Advertisements
Question
The glycosidic linkage present in maltose is ______.
Options
α, β-1, 2-glycosidic linkage
α-1, 4-glycosidic linkage
β-1, 4-glycosidic linkage
α-1, 6-glycosidic linkage
Advertisements
Solution
The glycosidic linkage present in maltose is α-1, 4-glycosidic linkage.
APPEARS IN
RELATED QUESTIONS
Give scientific reasons:
Hydrolysis of sucrose is called inversion.
Glucose on oxidation with dilute nitric acid gives _______________
Write the name of the polysaccharide used for the commercial preparation of glucose.
Draw the structure of α-D glucopyranose.
Write the name of the unit to which glucose unit is linked to from sucrose.
Write chemical reaction for following conversions
glucose into gluconic acid
Explain the classification of carbohydrates with examples.
What is monosaccharide?
Identify the given structure 'P' and 'Q'.

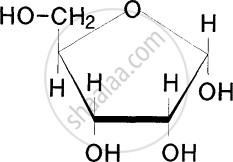
From the following which is a heteropolysaccharide?
Fructose is the fruit sugar and chemically it is ketohexose but it has a ______ rather than a ______.
From the following identify the materials that are made up of cellulose.
i. Plant cell wall
ii. Exoskeleton of arthropods
iii. Paper from plant pulp
iv. Cotton fibre
Identify the WRONG statement.
Match the Column I and Column II select the correct option.
| Column I | Column II | ||
| i. | Monosaccharides | a. | Stored energy in animals |
| ii. | Glycogen | b. | Polymer made from ß-glucose molecules |
| iii. | Cellulose | c. | Exoskeleton of arthropods |
| iv. | Chitin | d. | Building blocks of carbohydrates |
| e. | Plant hormone |
Chitin is a/an ______.
From the following identify the two types of glucose polymers present in starch.
Which among the following type of linkages is present in cellulose?
Prolonged heating of glucose with hot HI results in the formation of ____________.
Which of the following monosaccharide is a ketohexose?
Which one of the following is an oligosaccharide?
Which of the following statements is NOT true for glyceraldehyde?
Which of the following molecules reduces Fehling's solution?
What is the number of hydroxyl groups present in lactic acid?
What is the molecular formula of glyceraldehyde?
Identify the product Y in the following reaction.
\[\ce{Maltose ->[Hydrolysis] X ->[dil. HNO3][(excess)] Y}\]
How many moles of acetic acid are obtained in the reaction when one mole glucose is treated with excess acetic anhydride?
Which among the following is a product of hydrolysis of one mole raffinose?
How many hydroxyl groups are present in Erythrulose?
How many optical isomers are possible for a compound having four asymmetric carbon atoms?
Which one of the following rotates the plane polarized light towards left?
The correct corresponding order of names of four aldoses with configuration given below Respectively is:
\[\ce{Glucose ->[(HCN)] Product ->[(hydrolysis)] Product ->[(HI + Heat)] A}\], the compound A is:
Complete hydrolysis of cellulose gives ____________.
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Maltose
Which of the following groups contain polysaccharides?
If 'n' represents total number of asymmetric carbon atoms in a compound, then the possible· number of optical isomers of the compound is ______.
The two monosaccharides in a disaccharide are held together by ______ bonds.
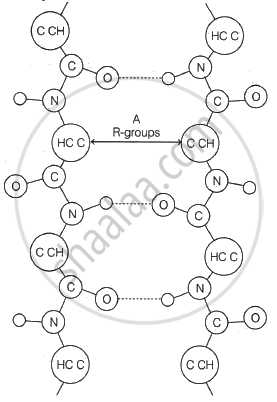
Match the Column I with Column II and choose the correct answer from options below:
| Column I | Column II |
| A. Purine | 1. Glycogen |
| B. Pyrimidine | 2. Cellulose |
| C. Structural polysaccharide | 3. Glucagon |
| D. Storage polysaccharide | 4. Adenine |
| 5. Cytosine |
Corn is immersed in boiling water. It is then cooled, and the solution becomes sweet. It is due to ______.
\[\ce{CH2OH - CO - (CHOH)4 - CH2OH}\] is an example of ______.
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active.
Assign D/L configuration to the following monosaccharides:
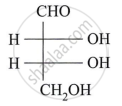
Carbohydrates that do not undergo hydrolysis further are called ______.
Why carbohydrates are generally optically active.
The linkage present in Lactose is ______.
Why are carbohydrates generally optically active?
Why carbohydrates are generally optically active?
\[\ce{CH2OH - CO - (CHOH)4 - CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active.
The sugar found in milk is ______.
