Advertisements
Advertisements
Question
The monomer of natural rubber is ______.
Options
Isoprene
Acrylonitrile
ε-Caprolactam
Tetrafluoroethylene
Advertisements
Solution
The monomer of natural rubber is isoprene.
Explanation:
Natural rubber : Monomer of natural rubber is isoprene (2-methyl - 1, 3-butadiene).
\[\begin{array}{cc}
\phantom{}\ce{CH3}\\
|\phantom{...}\\
\phantom{....}\ce{\underset{(isoprene)}{CH2 = C - CH = CH2}}\\
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Write the reactions involved in the preparation of PVC
The Zieglar-Natta catalyst is used in the preparation of _______.
(A) LDPE
(B) PHBV
(C) PAN
(D) HDPE
Write the structure of melamine.
Write the structures of the monomers used for getting the following polymers
Melamine – formaldehyde polymer
Write the monomers of the following polymer :

Choose the correct option from the given alternatives.
Which of the following is made up of polyamides?
Answer the following in one sentence.
Identify 'A' in the following reaction:
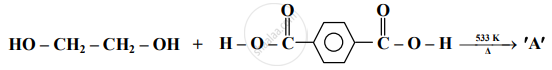
Answer the following in one sentence.
What type of intermolecular force leads to high-density polymer?
Answer the following in one sentence.
Identify thermoplastic and thermosetting plastic from the following:
- PET
- Urea formaldehyde resin
- Polythene
- Phenol formaldehyde resin
Answer the following.
Write the reaction of the formation of Nylon 6.
Draw the structures of polymers formed from the following monomers
\[\ce{n HOOC–R–COOH + n HO–R'–OH}\]
Answer the following.
Write name and formula of raw material from which bakelite is made.
Attempt the following:
Write the chemical reactions involved in the manufacture of Nylon 6,6.
Write the structure of isoprene and the polymer obtained from it.
Monomer used for preparation of polyacrylonitrile is _____________
Monomer of natural rubber is __________
Write a chemical reaction for the preparation of the following polymer.
polyacrylonitrile
Mention two uses of LDP.
Write the name of one example of each polymer in which following repeating units.
\[\begin{array}{cc}
\ce{(-CF2-CF2-), -[NH-(CH2)5-CO] -, -(CH2-CH-), (-CH2-CH2-)}\\
\phantom{............................}|\\
\phantom{..............................}\ce{CN}
\end{array}\]
Which among the following polymers is used for making handles of cooker?
Which of the following polymer is used in paints?
\[\ce{{n} CH2 = CH2 ->[333 K - 343 K][6 - 7 atm, catalyst] X}\]
Which of the following is CORRECT about polymer 'X'?
Which of the following polymers is obtained from chloroprene?
Which among the following polymers can NOT be remoulded?
Identify the monomers used in the preparation of Novolac.
Identify the catalyst used in the manufacture of high density polythene.
Which among the following monomers is used to prepare Teflon?
Which among the following polymers is an example of addition polymer?
Which among the following polymers is used to manufacture chemical containers?
Which among the following is an example of addition polymer?
Which of the following products is formed when benzaldehyde is treated with CH3MgBr and the addition product so obtained is subjected to acid hydrolysis?
F2C = CF2 is monomer of the polymer -
Trans - form of poly isoprene is:-
Which of the following is an example of polyester?
Polymer used in bullet proof glass is ______.
Which of the following polymer is used for manufacturing of buckets, dustbins, pipes, etc?
Which among the following polymers has high tensile strength and is used to obtain tyre cords?
Identify the monomer used to prepare neoprene.
Which of the foolowing polymer is used in the manufacture of insulators.
Answer the following.
Write the structure of isoprene and the polymer obtained from it.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the structure and name of the monomer of Nylon-6.
Name and draw the structure of the repeating unit in natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Which from following statements is NOT true regarding neoprene?
