Advertisements
Advertisements
Question
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2,8,1 |
2,8,7 |
2,5 |
1 |
Answer the following questions based on the table above:
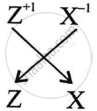
What is the formula of the compound formed between X and Z.
Advertisements
Solution
XZ is the formula of the compound formed between X and Z.
APPEARS IN
RELATED QUESTIONS
Define a chemical bond.
Match the atomic number 4, 8, 14, 15 and 19 with each of the following:
(a) A solid non-metal of valelncy 3.
(b) A gas of valency 2.
(c) A metal of valency 1.
(d) A non-metal of valency 4
What is the contribution of the following in atomic structure?
Democritus
Name the following:
Two atoms having the same number of protons and electrons but different number of neutrons.
Complete the following table relating to the atomic structure of some elements.
|
Element Symbol |
Atomic Number |
Mass Number |
Numbers of neutrons |
Number of Electrons |
Number of Protons |
|
Li |
3 |
6 |
|
|
|
|
Cl |
17 |
|
20 |
|
|
|
Na |
|
|
12 |
|
11 |
|
Al |
|
27 |
|
|
13 |
|
S |
|
32 |
16 |
|
|
Potassium (at No.19) and chlorine (at No.17) react to form a compound. Explain the formation of the compound on the basis of reduction.
Identify the following reaction as either oxidation or reduction:
Zn → Zn2+ + 2e-
Five atoms are labelled V to Z
| Atoms | Mass Number | Atomic Number |
| V | 40 | 20 |
| W | 19 | 9 |
| X | 7 | 3 |
| Y | 16 | 8 |
| Z | 14 | 7 |
- Which one of these atoms
(1) contains 7 protons;
(2) has an electronic configuration 2, 7? - Write down the formula of the compound formed by atoms X and Y.
Elements with stable electronic configurations have eight electrons in their valence shell. They are ______.
The element that would form cation due to the loss of electrons during the chemical reaction is ______.
