Advertisements
Advertisements
Question
An atom X with atomic number 20 combines with atom Y with atomic number 8. Draw the dot structure for the formation of the molecule XY.
Advertisements
Solution
Atom X is Calcium with atomic number 20 and atom Y is Oxygen with atomic number 8.
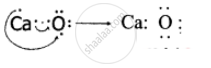
APPEARS IN
RELATED QUESTIONS
Match the atomic number 4, 8, 14, 15 and 19 with each of the following:
(a) A solid non-metal of valelncy 3.
(b) A gas of valency 2.
(c) A metal of valency 1.
(d) A non-metal of valency 4
(i) How many atoms of each kind are present in the following molecules: calcium oxide, chlorine, water, carbon tetrachloride?
(ii) How many electrons are required for their octet structure?
Fill in the blanks.
The electrovalent bond or ionic bond is called as _____ bond
Fill in the blank.
___________ single covalent bonds are formed between the carbon and chlorine atoms.
An element with the atomic number 19 will most likely combine chemically with the element whose atomic number is
What do you think would be the observation of `alpha` particle scattering experiment if carried out on (i) heavy nucleus like platinum (ii) light nuclei like litheum.
Element M forms a chloride with the formula MCl2 which is solid with a high melting point. What type of bond is in MCl2. Write the formula of the compound when M combines with sulphur, oxygen, and nitrogen.
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^23_11Na}\]
Draw the electron distribution diagram for the formation of Carbon di oxide (CO2 ) molecule.
How many atoms of each kind are present in the following molecule:
carbon tetrachloride
