Advertisements
Advertisements
प्रश्न
An atom X with atomic number 20 combines with atom Y with atomic number 8. Draw the dot structure for the formation of the molecule XY.
Advertisements
उत्तर
Atom X is Calcium with atomic number 20 and atom Y is Oxygen with atomic number 8.
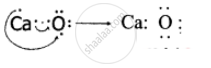
APPEARS IN
संबंधित प्रश्न
Give electron dot diagram of the following:
nitrogen
What type of bond is formed between two atoms, when the electronegative difference between them is zero?
How many atoms of each kind are present in the following molecules: calcium oxide, chlorine, water, carbon tetrachloride?
How many electrons are required by each atom mentioned in (a) to attain the nearest noble gas configuration?
How many atoms of each kind are present in the following molecule:
Calcium oxide
Draw orbit structure diagram of sodium chloride (NaCl).
An element M burns in oxygen to form an ionic compound MO. Write the formula of the compounds formed if this element is made to continue with chlorine and sulfur separately.
State the type of bonding in the following molecules.
Hydrogen chloride
Copy and complete the following table relating to the atomic structure of some elements:
| Element | Atomic Number | Mass Number | Number of protons | Number of Neutrons | Number of Electrons |
| Beryllium | 4 | 9 | |||
| Fluorine | 9 | 10 | |||
| Sodium | 12 | 11 | |||
| Aluminium | 27 | 13 | |||
| Phosphorus | 31 | 15 |
Complete the following table:
| a. | Dativebond | ______ |
| b. | CaH2 → Ca+H2 | ______ |
| c. | ______ | Reduction |
| d. | ______ | Redox reaction |
Elements A, B and C have atomic number 17, 19 and 10 respectively.
Write down the formula of the compound formed by two of the above elements.
