Advertisements
Advertisements
प्रश्न
An atom X with atomic number 20 combines with atom Y with atomic number 8. Draw the dot structure for the formation of the molecule XY.
Advertisements
उत्तर
Atom X is Calcium with atomic number 20 and atom Y is Oxygen with atomic number 8.
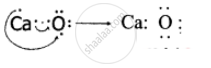
APPEARS IN
संबंधित प्रश्न
Mention the basic tendency of an atom which makes it to combine with other atoms.
Give electron dot diagram of the following:
Magnesium chloride
Explain the following briefly:
Helium does not form He2 molecule
What do you think would be the observation of `alpha` particle scattering experiment if carried out on (i) heavy nucleus like platinum (ii) light nuclei like litheum.
Define the term:
orbit
Five atoms are labeled A to E
|
Atoms |
Mass No |
Atomic No. |
|
A |
40 |
20 |
|
B |
19 |
9 |
|
C |
7 |
3 |
|
D |
16 |
8 |
|
E |
14 |
7 |
(a) Which one of these atoms:
(i) contains 7 protons
(ii) has electronic configuration 2,7
(b) Write down the formula in the compound formed between C and D
(c) Predict : (i) metals (ii) non-metals
Give a reason
Physical properties of isotopes are different.
Write down the electronic configuration of
(i) chlorine atom (ii) chlorine ion
Give the orbital diagram of the following:
Methane
How many atoms of each kind are present in the following molecule:
carbon tetrachloride
