Advertisements
Advertisements
प्रश्न
Complete the table give below.
| Element |
Atomic number |
Electron distribution | Valence electrons | Lewis dot structure |
| Lithium | 3 | 1s22s1 | ||
| Boron | 5 | 1s22s22p1 | ||
| Oxygen | 8 | 1s22s22p4 |
Advertisements
उत्तर
| Element |
Atomic number |
Electron distribution | Valence electrons | Lewis dot structure |
| Lithium | 3 | 1s22s1 | 1 | 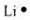 |
| Boron | 5 | 1s22s22p1 | 3 | 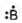 |
| Oxygen | 8 | 1s22s22p4 | 6 | 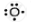 |
APPEARS IN
संबंधित प्रश्न
Give electron dot diagram of the following:
nitrogen
Give electron dot diagram of the following:
methane
Fill in the blanks.
The electrovalent bond or ionic bond is called as _____ bond
What do you think would be the observation of `alpha` particle scattering experiment if carried out on (i) heavy nucleus like platinum (ii) light nuclei like litheum.
Give a reason
Physical properties of isotopes are different.
Give the orbital diagram of the following:
Hydrogen chloride
Draw the electron distribution diagram for the formation of Carbon di oxide (CO2 ) molecule.
The element that would form cation due to the loss of electrons during the chemical reaction is ______.
Spot the error/correct the wrong statement:
In the formation of compounds, the inner shell electrons of an atom involved in bonding.
How many electrons are required or released by each atom in water molecule to attain the nearest noble gas configuration?
