Advertisements
Advertisements
Question
Complete the table give below.
| Element |
Atomic number |
Electron distribution | Valence electrons | Lewis dot structure |
| Lithium | 3 | 1s22s1 | ||
| Boron | 5 | 1s22s22p1 | ||
| Oxygen | 8 | 1s22s22p4 |
Advertisements
Solution
| Element |
Atomic number |
Electron distribution | Valence electrons | Lewis dot structure |
| Lithium | 3 | 1s22s1 | 1 | 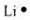 |
| Boron | 5 | 1s22s22p1 | 3 | 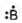 |
| Oxygen | 8 | 1s22s22p4 | 6 | 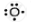 |
APPEARS IN
RELATED QUESTIONS
A Compound 'X' consists of only molecules. Hence 'X' will have
How many electrons are required or released by each atom in calcium oxide to attain the nearest noble gas configuration?
Why do `""_17^35"CI"` and `""_17^37"CI"` have the same chemical properties? In what respect do these atoms differ?
Explain fractional atomic mass. What is the fractional mass of chlorine?
Explain
Duplet rule for the formation of hydrogen
What do you understand by redox reactions?
In the formation of magnesium chloride (by direct combination between magnesium and chlorine), name the substance that is oxidized and the substance that is reduced.
What is a chemical bond?
Write the basic concept of Kossel – Lewis theory.
The one which is composed of all the three kinds of bond.[ionic, covalent and coordinate bond]
