Advertisements
Advertisements
प्रश्न
Complete the table give below.
| Element |
Atomic number |
Electron distribution | Valence electrons | Lewis dot structure |
| Lithium | 3 | 1s22s1 | ||
| Boron | 5 | 1s22s22p1 | ||
| Oxygen | 8 | 1s22s22p4 |
Advertisements
उत्तर
| Element |
Atomic number |
Electron distribution | Valence electrons | Lewis dot structure |
| Lithium | 3 | 1s22s1 | 1 | 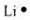 |
| Boron | 5 | 1s22s22p1 | 3 | 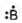 |
| Oxygen | 8 | 1s22s22p4 | 6 | 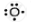 |
APPEARS IN
संबंधित प्रश्न
(i) How many atoms of each kind are present in the following molecules: calcium oxide, chlorine, water, carbon tetrachloride?
(ii) How many electrons are required for their octet structure?
Separate the following compounds into three categories - ionic, polar and covalent compounds; N2, NH4Cl, NH3, NO, NH4NO3, NCl3.
Match the column A with Column B.
|
Column A |
Column B |
|
(i) Sodium chloride (ii) Ammonium ion (iii) Carbon tetrachloride |
Covalent bond Ionic bond Covalent and coordinate bond |
How does the Modern atomic theory contradict and correlate with Dalton's atomic theory?
Five atoms are labeled A to E
|
Atoms |
Mass No |
Atomic No. |
|
A |
40 |
20 |
|
B |
19 |
9 |
|
C |
7 |
3 |
|
D |
16 |
8 |
|
E |
14 |
7 |
(a) Which one of these atoms:
(i) contains 7 protons
(ii) has electronic configuration 2,7
(b) Write down the formula in the compound formed between C and D
(c) Predict : (i) metals (ii) non-metals
Name the following:
Metal with valency 2
Draw orbit structure diagram of calcium oxide (CaO).
State the type of bonding in the following molecules.
Calcium oxide
Draw the geometric atomic structure of the three isotopes of hydrogen and the two isotopes of chlorine.
| Name of compound | Type of compound | |
| a. | Sodium chloride | ______ |
| b. | ______ | co-ordinate covalent compound |
| c. | Methane | ______ |
| d. | Fluorine molecule | ______ |
