Advertisements
Advertisements
प्रश्न
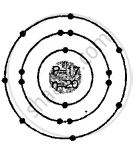
Draw the geometric atomic structure of the three isotopes of hydrogen and the two isotopes of chlorine.
Advertisements
उत्तर
Geometric atomic structures of three isotopes of hydrogen.

Each isotope has 1 electron, 1 proton.
But 0, 1, 2, neutrons respectively.
Two isotopes of chlorine
3517Cl

3717Cl
APPEARS IN
संबंधित प्रश्न
Define a chemical bond.
State differences between the properties of ionic compounds and covalent compounds.
How many atoms of each kind are present in the following molecules: calcium oxide, chlorine, water, carbon tetrachloride?
How many electrons are required by each atom mentioned in (a) to attain the nearest noble gas configuration?
How many atoms of each kind are present in the following molecule:
Calcium oxide
Why do they exist as monoatoms in molecules?
Elements with stable electronic configurations have eight electrons in their valence shell. They are ______.
Draw the electron distribution diagram for the formation of Carbon di oxide (CO2 ) molecule.
Match the following:
| 1. | Monoatomic gaseous atom | a. | Electrovalent bond |
| 2. | Octet rule | b. | Benzene |
| 3. | Ionic bond | c. | Water |
| 4. | Non-polar solvent | d. | Electronic theory of valence |
| 5. | Polar solvent | e. | Noble gases |
Choose the odd one out and write the reason:
How many atoms of each kind are present in the following molecule:
carbon tetrachloride
