Advertisements
Advertisements
प्रश्न
Draw the geometric atomic structure of the three isotopes of hydrogen and the two isotopes of chlorine.
Advertisements
उत्तर
Geometric atomic structures of three isotopes of hydrogen.

Each isotope has 1 electron, 1 proton.
But 0, 1, 2, neutrons respectively.
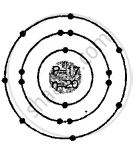
Two isotopes of chlorine
3517Cl

3717Cl
APPEARS IN
संबंधित प्रश्न
Define a chemical bond.
Elements A, B and C have atomic number 17, 19 and 10 respectively.
State which one is a non-metal.
Five atoms are labelled from A to E.
| Atoms | Mass No. | Atomic No. |
| A | 40 | 20 |
| B | 19 | 9 |
| C | 7 | 3 |
| D | 16 | 8 |
| E | 14 | 7 |
(a) Which one of these atoms:
(i) contains 7 protons,
(ii) has an electronic configuration 2, 7?
(b) Write down the formula of the compound formed between C and D.
(c) predict which are: (i) metals, (ii) non-metals?
An element with the atomic number 19 will most likely combine chemically with the element whose atomic number is
How many atoms of each kind are present in the following molecules: calcium oxide, chlorine, water, carbon tetrachloride?
How many electrons are required by each atom mentioned in (a) to attain the nearest noble gas configuration?
How many electrons are required or released by each atom in calcium oxide to attain the nearest noble gas configuration?
What do you understand by redox reactions? Explain oxidation and reduction in terms of loss or gain of electrons.
Give the orbital diagram of the following:
Hydrogen chloride
The element that would form cation due to the loss of electrons during the chemical reaction is ______.
How many atoms of each kind are present in the following molecule:
carbon tetrachloride
