Advertisements
Advertisements
Question
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2, 8, 1 |
2, 8, 7 |
2, 5 |
1 |
Answer the following question based on the table above:
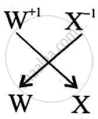
What is the formula of the compound formed between W and X.
Advertisements
Solution
WX is the formula of the compound formed between W and X.
APPEARS IN
RELATED QUESTIONS
Give electron dot diagram of the following:
nitrogen
In term of electron transfer, define
Reduction
What type of bond is formed between two atoms, when the electronegative difference between them is zero?
Separate the following compounds into three categories - ionic, polar and covalent compounds; N2, NH4Cl, NH3, NO, NH4NO3, NCl3.
Element X is a metal with a valency 2; Y is a non-metal with a valency 3.
(a) Write an equation to show how Y forms as ion
(b) If Y is a diatomic gas, write an equation for the direct combination of X and Y to form a compound.
Why do they exist as monoatoms in molecules?
Explain the following:
Carbon-12 and carbon-14 both show similar chemical properties.
Explain Octet rule with an example.
Complete the following table:
| Atoms | Atomic number | Electron distribution | |
| 1. | O | 8 | ______ |
| 2. | N | ______ | ______ |
| 3. | ______ | 17 | ______ |
| 4. | ______ | ______ | 2,8,2 |
How many atoms of each kind are present in the following molecule:
water
